Nitrobenzene gives N-phenylhydroxylamine by
A. Sn/HCl
B. \[{H_2}/Pd - C\]
C. Zn/NaOH
D. Zn/\[N{H_4}Cl\]
Answer
269.7k+ views
Hint: The conversion of nitrobenzene to N-phenylhydroxyamine is done with the help of a reducing agent. Here, the double-bonded oxygen group present in a nitro group is converted to oxime or a hydroxyl group.
Complete Step by Step Solution:
Nitrobenzene is converted to N-phenylhydroxylamine in presence of zinc dust along with ammonium chloride. It acts as a mild reducing agent which reduces the double bond oxygen present in the nitro group into an oxime. The zinc donates the electron pair to convert into a divalent cation.
The reaction between nitrobenzene and zinc dust in the presence of ammonium chloride is shown below.
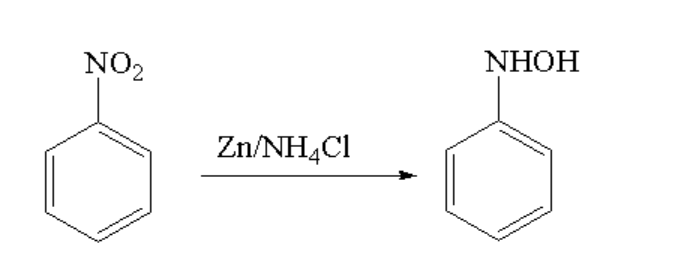
Image: Conversion of nitrobenzene to N-phenylhydroxyl amine
The reaction mechanism is shown below:
\[Ph - N{O_2} \to Ph - N = O \overset{2e^{-}/Zn^{2+}}{\rightarrow} PhNH(OH)\]
Here, Ph is the phenyl group
The nitrogen atom present in the nitro group is partially positive in nature. The zinc atom transfers its one electron to the nitrogen as a result one electron moves to oxygen and another electron to the nitrogen atom in \[N = O\]. The reaction of a proton with both \[ - {O^ - }\] forms N, N-hydroxybenzene which is very unstable due to the presence of two hydroxyl groups. By losing water molecules it forms the intermediate product nitrosobenzene which on reacting with zinc ion forms N-phenyl hydroxylamine.
Thus, Nitrobenzene gives N-phenylhydroxylamine by Zn/\[N{H_4}Cl\].
Therefore, the correct option is D.
Note: Reduction is defined as a reaction where there is a decrease in the oxidation state of the central atom. In the given reaction ammonium chloride is used as a promoter for zinc dust.
Complete Step by Step Solution:
Nitrobenzene is converted to N-phenylhydroxylamine in presence of zinc dust along with ammonium chloride. It acts as a mild reducing agent which reduces the double bond oxygen present in the nitro group into an oxime. The zinc donates the electron pair to convert into a divalent cation.
The reaction between nitrobenzene and zinc dust in the presence of ammonium chloride is shown below.

Image: Conversion of nitrobenzene to N-phenylhydroxyl amine
The reaction mechanism is shown below:
\[Ph - N{O_2} \to Ph - N = O \overset{2e^{-}/Zn^{2+}}{\rightarrow} PhNH(OH)\]
Here, Ph is the phenyl group
The nitrogen atom present in the nitro group is partially positive in nature. The zinc atom transfers its one electron to the nitrogen as a result one electron moves to oxygen and another electron to the nitrogen atom in \[N = O\]. The reaction of a proton with both \[ - {O^ - }\] forms N, N-hydroxybenzene which is very unstable due to the presence of two hydroxyl groups. By losing water molecules it forms the intermediate product nitrosobenzene which on reacting with zinc ion forms N-phenyl hydroxylamine.
Thus, Nitrobenzene gives N-phenylhydroxylamine by Zn/\[N{H_4}Cl\].
Therefore, the correct option is D.
Note: Reduction is defined as a reaction where there is a decrease in the oxidation state of the central atom. In the given reaction ammonium chloride is used as a promoter for zinc dust.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Understanding Atomic Structure for Beginners

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




