Molecular weight of protein is:
A. > 12000
B. < 6000
C. < 12000
D. 600 – 3000
Answer
249.6k+ views
Hint: As protein made up of a hundred to a thousand of smaller structural units which is called amino acid. There are majorly 20 amino acids which are arranged in different manners to generate a protein. Molecular weight of proteins can be calculated by sum of individual weight of all amino acids which are used to build the protein.
Complete Step by Step Solution:
Amino acid, its name itself suggesting that amino acid is a combination of amino group and acid (carboxylic acid) such as
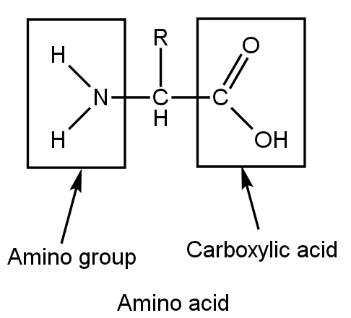
This type of amino acid combines with one or more other amino acids through peptide linkage. If there is less number of peptide linkage then it is called oligopeptides whereas if large number of Question:
For proteins which are polypeptides will consist 100 to 1000 of amino acids which together give average molecular weight greater than 1000 to 10,000 and can cross a million value. For sake of convenience, 110 Dalton or 110 amu is the average weight of single amino acid. After multiplying this value of weight to the total number of amino acids present in protein, give the average molecular weight of that protein.
So, molecular weight of protein must be greater than 12,000, thus, the correct option is A.
Note: It is important to note that we can calculate average molecular weight of protein because of the presence of many other variable groups along with carbon, nitrogen, carbonyl group, hydrogen and Sulphur and also due to the complexity of polypeptides.
Complete Step by Step Solution:
Amino acid, its name itself suggesting that amino acid is a combination of amino group and acid (carboxylic acid) such as

This type of amino acid combines with one or more other amino acids through peptide linkage. If there is less number of peptide linkage then it is called oligopeptides whereas if large number of Question:
For proteins which are polypeptides will consist 100 to 1000 of amino acids which together give average molecular weight greater than 1000 to 10,000 and can cross a million value. For sake of convenience, 110 Dalton or 110 amu is the average weight of single amino acid. After multiplying this value of weight to the total number of amino acids present in protein, give the average molecular weight of that protein.
So, molecular weight of protein must be greater than 12,000, thus, the correct option is A.
Note: It is important to note that we can calculate average molecular weight of protein because of the presence of many other variable groups along with carbon, nitrogen, carbonyl group, hydrogen and Sulphur and also due to the complexity of polypeptides.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




