Isopropyl alcohol is obtained by reacting which of the following alkenes with conc. \[{{\rm{H}}_{\rm{2}}}{\rm{S}}{{\rm{O}}_{\rm{4}}}\] and \[{{\rm{H}}_{\rm{2}}}{\rm{O}}\]
A) Ethylene
B) Propylene
C) 2-methylpropene
D) Isoprene
Answer
257.4k+ views
Hint: The chemical representation of the compound isopropyl alcohol is \[{\rm{C}}{{\rm{H}}_{\rm{3}}} - {\rm{CH(OH)}} - {\rm{C}}{{\rm{H}}_{\rm{3}}}\] . Now, we have to identify the alkene that gives isopropyl alcohol on reaction with conc. Sulphuric acid.
Complete Step by Step Answer:
Let’s discuss the options one by one.
Option A is ethylene (Ethene). The chemical representation of ethene is \[{\rm{C}}{{\rm{H}}_{\rm{2}}} = {\rm{C}}{{\rm{H}}_{\rm{2}}}\] . But, in the isopropyl alcohol, there is the presence of three atoms of carbon. So, A does not give isopropyl alcohol when it reacts with conc. sulphuric acid.
Option C is 2-methyl propene. The chemical representation of 2-methyl propene is \[{{\rm{H}}_{\rm{3}}}{\rm{C}} - {\rm{HC(C}}{{\rm{H}}_{\rm{3}}}{\rm{)}} - {\rm{C}}{{\rm{H}}_{\rm{3}}}\] . So, there is a methyl substituent in C2. But, in isopropyl alcohol, there is no methyl substituent. Therefore, 2-methylpropene also does not give isopropyl alcohol.
Option D is isoprene. Isoprene has the chemical representation of \[{\rm{C}}{{\rm{H}}_{\rm{2}}} = {\rm{C(C}}{{\rm{H}}_{\rm{3}}}{\rm{)}} - {\rm{CH}} = {\rm{C}}{{\rm{H}}_{\rm{2}}}\] . It has five atoms of carbon in the compound. So, it does not give isopropyl alcohol when it undergoes a reaction with concentrated sulphuric acid.
Option B is propylene. The reaction of propylene with conc. Sulphuric acid gives isopropyl alcohol. The reaction happens in the following way:
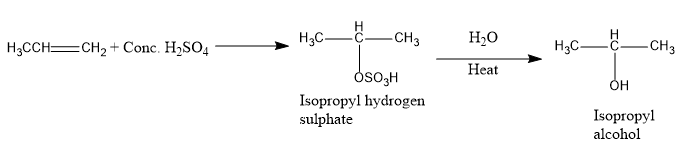
Image: Formation of isopropyl alcohol
Therefore, when propylene undergoes a reaction with sulphuric acid, isopropyl alcohol forms.
Therefore, option B is right.
Note: Isopropyl alcohol is a liquid of no colour and is a useful component in many household items namely disinfectants, cleaners, hand sanitizers, etc. Its use in the pharmaceutical industry is also significant.
Complete Step by Step Answer:
Let’s discuss the options one by one.
Option A is ethylene (Ethene). The chemical representation of ethene is \[{\rm{C}}{{\rm{H}}_{\rm{2}}} = {\rm{C}}{{\rm{H}}_{\rm{2}}}\] . But, in the isopropyl alcohol, there is the presence of three atoms of carbon. So, A does not give isopropyl alcohol when it reacts with conc. sulphuric acid.
Option C is 2-methyl propene. The chemical representation of 2-methyl propene is \[{{\rm{H}}_{\rm{3}}}{\rm{C}} - {\rm{HC(C}}{{\rm{H}}_{\rm{3}}}{\rm{)}} - {\rm{C}}{{\rm{H}}_{\rm{3}}}\] . So, there is a methyl substituent in C2. But, in isopropyl alcohol, there is no methyl substituent. Therefore, 2-methylpropene also does not give isopropyl alcohol.
Option D is isoprene. Isoprene has the chemical representation of \[{\rm{C}}{{\rm{H}}_{\rm{2}}} = {\rm{C(C}}{{\rm{H}}_{\rm{3}}}{\rm{)}} - {\rm{CH}} = {\rm{C}}{{\rm{H}}_{\rm{2}}}\] . It has five atoms of carbon in the compound. So, it does not give isopropyl alcohol when it undergoes a reaction with concentrated sulphuric acid.
Option B is propylene. The reaction of propylene with conc. Sulphuric acid gives isopropyl alcohol. The reaction happens in the following way:

Image: Formation of isopropyl alcohol
Therefore, when propylene undergoes a reaction with sulphuric acid, isopropyl alcohol forms.
Therefore, option B is right.
Note: Isopropyl alcohol is a liquid of no colour and is a useful component in many household items namely disinfectants, cleaners, hand sanitizers, etc. Its use in the pharmaceutical industry is also significant.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




