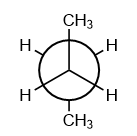
In the given conformation, ${C_2}$ is rotated about ${C_2} - {C_3}$ bond anticlockwise by an angle of ${120^o}$, then the conformation obtained is:
(A) Fully eclipsed conformation
(B) Partially eclipsed conformation
(C) Gauche conformation
(D) Staggered conformation
Answer
249.9k+ views
Hint: The conformation discussed in the question is of Newman projection which visualises the chemical bond conformation from front to back with respect to the relative position of groups attached to the two carbon atoms. The carbon at the front which is represented by a line is known as proximal while the carbon atom at the back which is represented by a circle is known as distal.
Complete Step by Step Solution:
Before comparing the stages of different conformations, let's discuss the different types of conformations shown in Newman projection as follows:
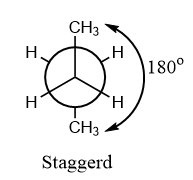
Staggered conformation: When all the groups of different axes in the conformation are at an angle of ${180^o}$ to each other, then the conformation is known as staggered conformation and it is the most stable conformation among all the conformations formed.
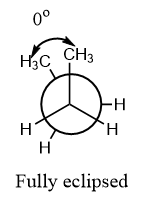
Fully eclipsed: When there is an angle of ${0^o}$ between the similar groups on different axes, i.e., the groups overlap each other, then the conformation is known as a fully eclipsed conformation. It is the most unstable conformation because a repulsion is experienced by the atoms when they overlap.
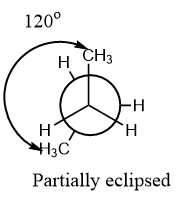
Partially eclipsed: When there is an angle of ${120^o}$ between the similar groups on different axes then the conformation is known as a partially eclipsed conformation.
Gauche conformation: This is a special kind of conformation in which there is an angle of ${60^o}$ between the similar groups on different axes. The Gauche conformation is unstable than the staggered conformation due to the steric hindrance between the two methyl groups but is comparatively stable than the partially and fully eclipsed conformations.
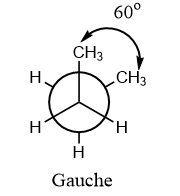
In the given question, the compound is given in staggered conformation and as per the given rotation, the following conformation will be formed:
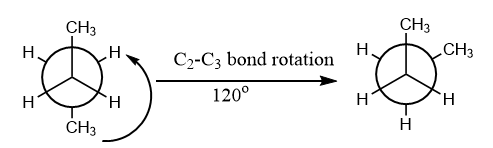
Hence, the formation of gauche conformation will take place on an anti-clock rotation of ${C_2}$ about ${C_2} - {C_3}$ bond by ${120^o}$. Therefore, option (C) is the correct answer.
Note: There are 2 special types of angular momentum of an object: the spin angular momentum is the angular momentum about the object centre of mass while the orbital angular momentum is the angular momentum about the chosen centre of rotation.
Complete Step by Step Solution:
Before comparing the stages of different conformations, let's discuss the different types of conformations shown in Newman projection as follows:
Staggered conformation: When all the groups of different axes in the conformation are at an angle of ${180^o}$ to each other, then the conformation is known as staggered conformation and it is the most stable conformation among all the conformations formed.

Fully eclipsed: When there is an angle of ${0^o}$ between the similar groups on different axes, i.e., the groups overlap each other, then the conformation is known as a fully eclipsed conformation. It is the most unstable conformation because a repulsion is experienced by the atoms when they overlap.

Partially eclipsed: When there is an angle of ${120^o}$ between the similar groups on different axes then the conformation is known as a partially eclipsed conformation.

Gauche conformation: This is a special kind of conformation in which there is an angle of ${60^o}$ between the similar groups on different axes. The Gauche conformation is unstable than the staggered conformation due to the steric hindrance between the two methyl groups but is comparatively stable than the partially and fully eclipsed conformations.

In the given question, the compound is given in staggered conformation and as per the given rotation, the following conformation will be formed:

Hence, the formation of gauche conformation will take place on an anti-clock rotation of ${C_2}$ about ${C_2} - {C_3}$ bond by ${120^o}$. Therefore, option (C) is the correct answer.
Note: There are 2 special types of angular momentum of an object: the spin angular momentum is the angular momentum about the object centre of mass while the orbital angular momentum is the angular momentum about the chosen centre of rotation.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




