In presence of \[AlC{l_3}\], benzene and n - propyl bromide react in Friedel - Craft's reaction to form:
A. N-propyl benzene
B. 1,2-Dinormal propyl benzene
C. 1,4-Dinormal propyl benzene
D. Isopropyl benzene
Answer
262.8k+ views
Hint: Friedel-Crafts reaction in consideration here is an alkylation reaction. It proceeds via an electrophilic aromatic substitution mechanism where the n-propyl bromide provides the electrophile intermediate.
Complete Step by Step Solution:
Friedel-Crafts reactions are a set of reactions which involve the attaching of a substituent to an aromatic ring. Friedel-Crafts reactions are of two types, alkylation reactions, in which an alkyl group (\[ - R\]) gets attached to a ring, and acylation reactions involving the attaching of an acyl group (\[ - RC = O\]) to a ring. In this reaction, the catalyst employed is aluminium chloride (\[AlC{l_3}\]) which is a strong Lewis acid. The alkylating agents have traditionally been alkyl halides.
Let’s have a look at how benzene and n-propyl bromide undergo Friedel-Craft’s reaction in presence of aluminium chloride.
In the first step, the n-propyl bromide loses its bromine atom with the help of aluminium chloride as shown. Since aluminium chloride is a strong Lewis acid, it accepts electrons from the bromine atom of n-propyl bromide, and it leads to the formation of an n-propyl carbocation.
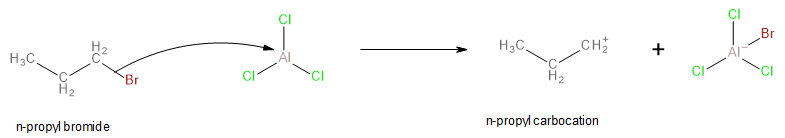
Image: Formation of n-propyl carbocation
The n-propyl carbocation formed is primary. We know that primary carbocations are the least stable (order of stability of carbocations: \[1^\circ < 2^\circ < 3^\circ \]). Since secondary carbocations are more stable, if the n-propyl carbocation can somehow become a secondary carbocation, it would become more stable. The n-propyl carbocation does indeed turn into a secondary carbocation through the rearrangement of an alpha-hydrogen. This process is called a 1,2-hydride shift since the hydrogen rearranges as a hydride and it occurs from the alpha-carbon (locant 2) to the cationic carbon (locant 1).
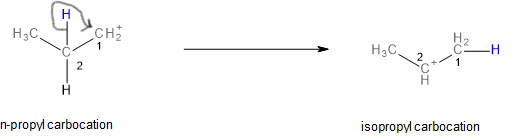
Image: Rearrangement into a more stable, secondary carbocation
The product is a more stable, secondary, isopropyl carbocation.
The isopropyl carbocation is then attacked by a \[\pi \]electron pair of benzene followed by the liberation of hydrogen bromide (\[HBr\]) to form isopropyl benzene which is commonly known as cumene.
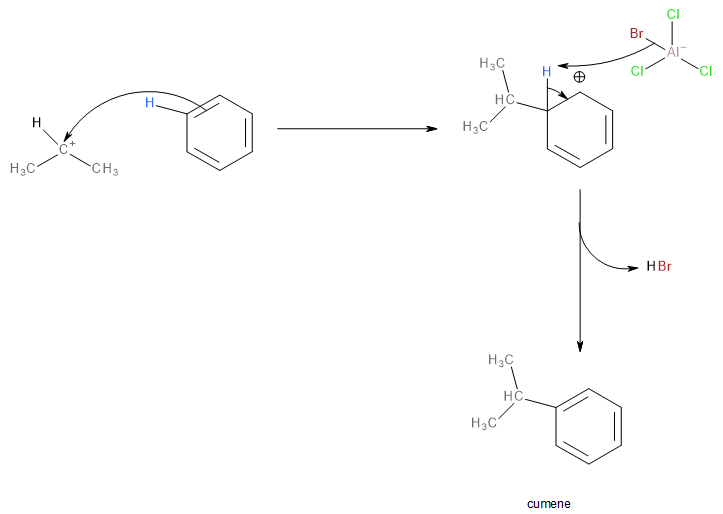
Image: Nucleophilic attack of a benzene ring on the carbocation.
Thus, the correct option is D.
Note: The rearrangement of the primary n-propyl carbocation into a more stable, secondary, isopropyl carbocation via 1,2-hydride shift is a very crucial step in this reaction. This is where quite a few students may go wrong. If they are unaware of this rearrangement step, they might mark option A as the answer to this question. They would be wrong.
Complete Step by Step Solution:
Friedel-Crafts reactions are a set of reactions which involve the attaching of a substituent to an aromatic ring. Friedel-Crafts reactions are of two types, alkylation reactions, in which an alkyl group (\[ - R\]) gets attached to a ring, and acylation reactions involving the attaching of an acyl group (\[ - RC = O\]) to a ring. In this reaction, the catalyst employed is aluminium chloride (\[AlC{l_3}\]) which is a strong Lewis acid. The alkylating agents have traditionally been alkyl halides.
Let’s have a look at how benzene and n-propyl bromide undergo Friedel-Craft’s reaction in presence of aluminium chloride.
In the first step, the n-propyl bromide loses its bromine atom with the help of aluminium chloride as shown. Since aluminium chloride is a strong Lewis acid, it accepts electrons from the bromine atom of n-propyl bromide, and it leads to the formation of an n-propyl carbocation.

Image: Formation of n-propyl carbocation
The n-propyl carbocation formed is primary. We know that primary carbocations are the least stable (order of stability of carbocations: \[1^\circ < 2^\circ < 3^\circ \]). Since secondary carbocations are more stable, if the n-propyl carbocation can somehow become a secondary carbocation, it would become more stable. The n-propyl carbocation does indeed turn into a secondary carbocation through the rearrangement of an alpha-hydrogen. This process is called a 1,2-hydride shift since the hydrogen rearranges as a hydride and it occurs from the alpha-carbon (locant 2) to the cationic carbon (locant 1).

Image: Rearrangement into a more stable, secondary carbocation
The product is a more stable, secondary, isopropyl carbocation.
The isopropyl carbocation is then attacked by a \[\pi \]electron pair of benzene followed by the liberation of hydrogen bromide (\[HBr\]) to form isopropyl benzene which is commonly known as cumene.

Image: Nucleophilic attack of a benzene ring on the carbocation.
Thus, the correct option is D.
Note: The rearrangement of the primary n-propyl carbocation into a more stable, secondary, isopropyl carbocation via 1,2-hydride shift is a very crucial step in this reaction. This is where quite a few students may go wrong. If they are unaware of this rearrangement step, they might mark option A as the answer to this question. They would be wrong.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




