H-O-H bond angle in \[{{H}_{2}}O\] is \[104.5{}^\circ \] and not \[109{}^\circ 28'\] because of
(A) Lone pair-lone pair repulsion
(B) Lone pair-bond pair repulsion
(C) Bond pair-bond pair repulsion
(D) High electronegativity of oxygen
Answer
263.4k+ views
Hint: VSEPR (Valence shell electron repulsion theory) explains the minimal variations we can see from the theoretical bond angles in some of the molecules. As the name suggests it involves repulsion between valence shell orbitals.
Complete step by step solution:
This is the shape of the Water molecule.
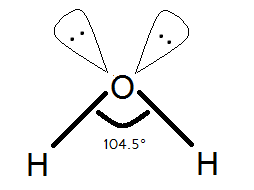
We know and we can see that oxygen has two lone pair on itself. The water molecule has \[s{{p}^{3}}\] hybridization that means it is supposed to have bond angles of \[109{}^\circ 28'\] but there is a special case.
In terms of repulsion, lone pair-lone pair repulsion is higher than any other repulsion.
So, bond pair-bond pair repulsion and lone pair-bond pair repulsion is lower than lone pair- lone pair repulsion.
So, due to lone pair-lone pair repulsion of two lone pairs of the oxygen atom, they both tend to repulse each other and try to stay as far as possible to each other. In that case, the bond angles of \[s{{p}^{3}}\]hybridization do not remain same and have less angle due to that because there is an increase in the angle between two lone pairs and hence there is a decrease in H-O-H angle.
So, that is the reason why Water has H-O-H bond angle of \[104.5{}^\circ \] and not \[109{}^\circ 28'\].
So, the correct answer is (A) Lone pair-lone pair repulsion.
Additional information:
Below are some of the important postulates of VSEPR theory.
-The shape of the molecules depends upon the number of valence cell electron pairs around the central atom.
-Pairs of electrons in valence shell repel each other since electron cloud is negatively charged.
-These electron pairs tend to arrange is space in a way that minimum repulsion is there.
Note: Remember that oxygen atom has two lone pairs in the structure of water molecule. Do not forget to consider the lone pair while predicting the shape or bond angles of a molecule.
Complete step by step solution:
This is the shape of the Water molecule.

We know and we can see that oxygen has two lone pair on itself. The water molecule has \[s{{p}^{3}}\] hybridization that means it is supposed to have bond angles of \[109{}^\circ 28'\] but there is a special case.
In terms of repulsion, lone pair-lone pair repulsion is higher than any other repulsion.
So, bond pair-bond pair repulsion and lone pair-bond pair repulsion is lower than lone pair- lone pair repulsion.
So, due to lone pair-lone pair repulsion of two lone pairs of the oxygen atom, they both tend to repulse each other and try to stay as far as possible to each other. In that case, the bond angles of \[s{{p}^{3}}\]hybridization do not remain same and have less angle due to that because there is an increase in the angle between two lone pairs and hence there is a decrease in H-O-H angle.
So, that is the reason why Water has H-O-H bond angle of \[104.5{}^\circ \] and not \[109{}^\circ 28'\].
So, the correct answer is (A) Lone pair-lone pair repulsion.
Additional information:
Below are some of the important postulates of VSEPR theory.
-The shape of the molecules depends upon the number of valence cell electron pairs around the central atom.
-Pairs of electrons in valence shell repel each other since electron cloud is negatively charged.
-These electron pairs tend to arrange is space in a way that minimum repulsion is there.
Note: Remember that oxygen atom has two lone pairs in the structure of water molecule. Do not forget to consider the lone pair while predicting the shape or bond angles of a molecule.
Recently Updated Pages
Area of an Octagon Formula Explained Simply

Absolute Pressure Formula Explained: Key Equation & Examples

Difference Between Atom and Molecule: JEE Main 2026

Difference Between Vapor and Gas: JEE Main 2026

Carbon Dioxide Formula - Definition, Uses and FAQs

Central Angle of a Circle Formula Explained Quickly

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




