\[{H_2}S{O_5} + {H_2}O \to {H_2}S{O_4} + {H_2}{O_2}\]
Oxidation number of sulphur in ${{\text{H}}_{\text{2}}}{\text{S}}{{\text{O}}_{\text{5}}}$ in the above reaction is:
a) 6
b) 4
c) 5
d) 2
Answer
262.8k+ views
Hint: You need to know the structure of ${{\text{H}}_{\text{2}}}{\text{S}}{{\text{O}}_{\text{5}}}$ to solve this problem. Because linkages of oxygen may be different in this structure. Calculate the total charges and equate them to zero to find out the oxidation number of sulphur.
Complete step by step answer:
We need to know the structure of the ${{\text{H}}_{\text{2}}}{\text{S}}{{\text{O}}_{\text{5}}}$ molecule. So, let us look at the structure of ${{\text{H}}_{\text{2}}}{\text{S}}{{\text{O}}_{\text{5}}}$ molecule.
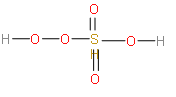
Now let us calculate the oxidation number of sulphur in this structure. Let us take the oxidation number of sulphur as x.
The charges of oxygen in the peroxide linkages are -1 and others are -2.
So, $2\left( { + 1} \right) + 2\left( { - 1} \right) + 3\left( { - 2} \right) + x = 0$
From this, $x = + 6$
Therefore, the oxidation number of sulphur in ${{\text{H}}_{\text{2}}}{\text{S}}{{\text{O}}_{\text{5}}}$molecule is +6.
So, the correct option is (a).
Additional Information:
Oxidation number is also called oxidation state, it is the total number of electrons that an atom either gains or loses in order to form a chemical bond with another atom.
An oxidation number is a number that is assigned to an atom in a substance. The oxidation number could be positive, negative or zero, and it indicates if electrons are lost or gained. Also, we can say that oxidation number is a number that helps us keep track of electrons in an atom.
Note: You should not be confused with oxidation number and valency. Valency cannot be negative. But oxidation numbers can be negative. Also, please note that the charge on the oxygen in peroxide linkage is -1 and in normal oxygen linkage, it is -2.
Complete step by step answer:
We need to know the structure of the ${{\text{H}}_{\text{2}}}{\text{S}}{{\text{O}}_{\text{5}}}$ molecule. So, let us look at the structure of ${{\text{H}}_{\text{2}}}{\text{S}}{{\text{O}}_{\text{5}}}$ molecule.

Now let us calculate the oxidation number of sulphur in this structure. Let us take the oxidation number of sulphur as x.
The charges of oxygen in the peroxide linkages are -1 and others are -2.
So, $2\left( { + 1} \right) + 2\left( { - 1} \right) + 3\left( { - 2} \right) + x = 0$
From this, $x = + 6$
Therefore, the oxidation number of sulphur in ${{\text{H}}_{\text{2}}}{\text{S}}{{\text{O}}_{\text{5}}}$molecule is +6.
So, the correct option is (a).
Additional Information:
Oxidation number is also called oxidation state, it is the total number of electrons that an atom either gains or loses in order to form a chemical bond with another atom.
An oxidation number is a number that is assigned to an atom in a substance. The oxidation number could be positive, negative or zero, and it indicates if electrons are lost or gained. Also, we can say that oxidation number is a number that helps us keep track of electrons in an atom.
Note: You should not be confused with oxidation number and valency. Valency cannot be negative. But oxidation numbers can be negative. Also, please note that the charge on the oxygen in peroxide linkage is -1 and in normal oxygen linkage, it is -2.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




