Give the names of the following compounds.
(i) \[HClO\]
(ii) \[HCl{O_2}\]
(iii) \[HCl{O_3}\]
(iv) \[HCl{O_4}\]
Answer
255.9k+ views
Hint: Chemical name is used to represent any chemical compound. For different chemical compounds, different unique chemical names are applied.
Complete step-by-step answer:
Acids are chemical compounds with a pH of less than 7. Acids generally dissociate when added to water to give hydrogen ions.
(i) \[HClO\]
The structure is shown below.
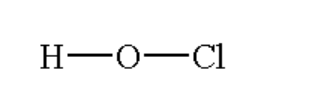
Image: \[HClO\]
Here, one oxygen atom is bonded to one hydrogen atom and one chlorine atom.
The chemical name of \[HClO\]is hypochlorous acid. It is a weak acid.
(ii) \[HCl{O_2}\]
The structure is shown below.
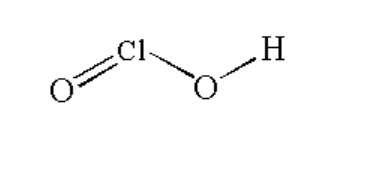
Image: \[HCl{O_2}\]
Here, the chlorine atom is bonded with an oxygen atom by a double bond and one oxygen atom by a single bond and one oxygen atom is bonded with hydrogen by a single bond.
The chemical name of \[HCl{O_2}\]chlorous acid. It is a oxoacid of chlorine.
(iii) \[HCl{O_3}\]
The structure is shown below.
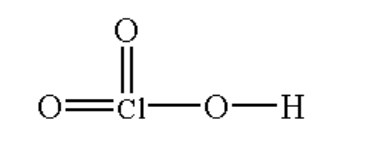
Image: \[HCl{O_3}\]
Here the chlorine atom is bonded with two oxygen atoms by a double bond and one oxygen atom by a single bond and the oxygen atom is bonded with hydrogen by a single bond.
The chemical name of \[HCl{O_3}\] is chloric acid.It is a colourless liquid.
(iv) \[HCl{O_4}\]
The structure is shown below.
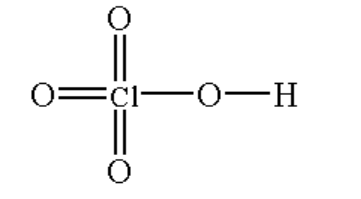
Image: \[HCl{O_4}\]
Here, the chlorine atom is bonded with three oxygen atoms by a double bond and one oxygen atom by a single bond and the oxygen atom is bonded with hydrogen by a single bond.
The chemical name of \[HCl{O_4}\]is perchloric acid. It is a colourless, odourless solution.
Note: The given chemical compounds, hypochlorous acid, chlorous acid, chloric acid, and perchloric acid come under chlorine acid. Hydrochloric acid also come under chlorine acid.
Complete step-by-step answer:
Acids are chemical compounds with a pH of less than 7. Acids generally dissociate when added to water to give hydrogen ions.
(i) \[HClO\]
The structure is shown below.
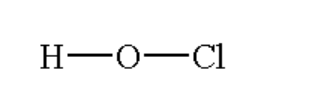
Image: \[HClO\]
Here, one oxygen atom is bonded to one hydrogen atom and one chlorine atom.
The chemical name of \[HClO\]is hypochlorous acid. It is a weak acid.
(ii) \[HCl{O_2}\]
The structure is shown below.

Image: \[HCl{O_2}\]
Here, the chlorine atom is bonded with an oxygen atom by a double bond and one oxygen atom by a single bond and one oxygen atom is bonded with hydrogen by a single bond.
The chemical name of \[HCl{O_2}\]chlorous acid. It is a oxoacid of chlorine.
(iii) \[HCl{O_3}\]
The structure is shown below.
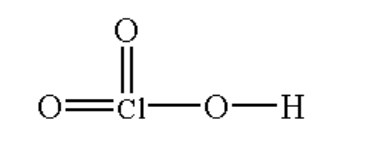
Image: \[HCl{O_3}\]
Here the chlorine atom is bonded with two oxygen atoms by a double bond and one oxygen atom by a single bond and the oxygen atom is bonded with hydrogen by a single bond.
The chemical name of \[HCl{O_3}\] is chloric acid.It is a colourless liquid.
(iv) \[HCl{O_4}\]
The structure is shown below.

Image: \[HCl{O_4}\]
Here, the chlorine atom is bonded with three oxygen atoms by a double bond and one oxygen atom by a single bond and the oxygen atom is bonded with hydrogen by a single bond.
The chemical name of \[HCl{O_4}\]is perchloric acid. It is a colourless, odourless solution.
Note: The given chemical compounds, hypochlorous acid, chlorous acid, chloric acid, and perchloric acid come under chlorine acid. Hydrochloric acid also come under chlorine acid.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




