Formal charge on nitrogen and oxygen in $NO_3^ - $ ion are respectively:
(A) 0, $ - \dfrac{1}{3}$
(B) +1, $ - \dfrac{1}{3}$
(C) +1, $ - \dfrac{2}{3}$
(D) 0, $ - \dfrac{2}{3}$
Answer
265.5k+ views
Hint: To answer this question we must first recall the concept of a formal charge. We would also have to remember how it was calculated via formula and by instinct to some extent.
Complete step by step solution:
-Formal charge is actually the charge assigned to an atom, under the assumption that the electrons in a chemical bond are shared equally between the atoms and that they have almost no difference in their electronegativities.
-To calculate formal charges, we assign electrons in the molecule to individual atoms according to these rules:
(1)Non-bonding electrons are only assigned to the atom on which they are located.
(2)Bonding electrons are divided equally between the two bonded atoms, so one electron from each bond goes to each atom.
-On drawing the structure of nitrite ion, we can predict the number of electrons each element entails.
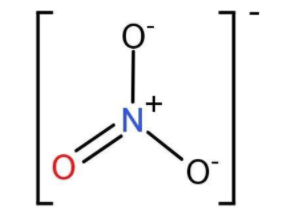
-The mathematical formula to calculate formal charge is:
$F.C. = V - N - \dfrac{B}{2}$
Where V = number of valence electrons;
N = number of nonbonding electrons;
B = Number of electrons in a covalent bond.
-For Nitrogen:
From the structure itself, we find a positive charge on Nitrogen, hence it carries a formal charge of +1. This is what we assign to every element of a compound. We are counting 4 electrons (4 bonding electrons, no nonbonding electrons). Since N is in Group 15, we have one less electron than N should have, and so it has a +1 charge.
-For oxygen,
From the structure, we see that, for the O with the double bond, we are counting 6 electrons for it (4 nonbonding electrons and 2 bonding electrons (one per band). Since O is in Group 16, that’s how many electrons it should have, so the charge for that O is zero. For either of the O atoms with a single bond, we can count 7 electrons (6 nonbonding electrons and one bonding electron). That’s one more than 6, so those O atoms have a charge of −1 each. Hence for 3 O atoms, we find the formal charge is $ - \dfrac{1}{3}$.
Hence the correct answer is Option (B) +1, $ - \dfrac{1}{3}$.
Additional information:
The structure with the most formal charges of zero on atoms is the most stable Lewis structure. In cases where there are positive or negative formal charges on various atoms, the most stable structures generally have negative formal charges on the more electronegative atoms and positive formal charges on the less electronegative atoms.
Note: The single bound oxygen atoms have nine electrons linked with them moreover they have a negative charge overall. This means the nitrate ion has an overall charge of -1.
Complete step by step solution:
-Formal charge is actually the charge assigned to an atom, under the assumption that the electrons in a chemical bond are shared equally between the atoms and that they have almost no difference in their electronegativities.
-To calculate formal charges, we assign electrons in the molecule to individual atoms according to these rules:
(1)Non-bonding electrons are only assigned to the atom on which they are located.
(2)Bonding electrons are divided equally between the two bonded atoms, so one electron from each bond goes to each atom.
-On drawing the structure of nitrite ion, we can predict the number of electrons each element entails.

-The mathematical formula to calculate formal charge is:
$F.C. = V - N - \dfrac{B}{2}$
Where V = number of valence electrons;
N = number of nonbonding electrons;
B = Number of electrons in a covalent bond.
-For Nitrogen:
From the structure itself, we find a positive charge on Nitrogen, hence it carries a formal charge of +1. This is what we assign to every element of a compound. We are counting 4 electrons (4 bonding electrons, no nonbonding electrons). Since N is in Group 15, we have one less electron than N should have, and so it has a +1 charge.
-For oxygen,
From the structure, we see that, for the O with the double bond, we are counting 6 electrons for it (4 nonbonding electrons and 2 bonding electrons (one per band). Since O is in Group 16, that’s how many electrons it should have, so the charge for that O is zero. For either of the O atoms with a single bond, we can count 7 electrons (6 nonbonding electrons and one bonding electron). That’s one more than 6, so those O atoms have a charge of −1 each. Hence for 3 O atoms, we find the formal charge is $ - \dfrac{1}{3}$.
Hence the correct answer is Option (B) +1, $ - \dfrac{1}{3}$.
Additional information:
The structure with the most formal charges of zero on atoms is the most stable Lewis structure. In cases where there are positive or negative formal charges on various atoms, the most stable structures generally have negative formal charges on the more electronegative atoms and positive formal charges on the less electronegative atoms.
Note: The single bound oxygen atoms have nine electrons linked with them moreover they have a negative charge overall. This means the nitrate ion has an overall charge of -1.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




