For an ideal gas, in an isothermal process
A. Heat content remains constant
B. Heat content and temperature remains constant
C. Temperature remains constant
D. None of the above
Answer
255k+ views
Hint: In this problem, to determine which option is correct for an isothermal thermodynamic process, we must know the basic fundamentals and applications of an isothermal process following the condition $\Delta T = 0$ hence, use this condition to state the answer for the given situation with proper explanation.
Complete step by step solution:
The term "isothermal process" refers to a substance, an item, or a system changing at a specific constant temperature. Isothermal Process in thermodynamics is a process during which the temperature $T$ of a system remains constant whereas other variables like pressure $P$ and volume $V$ may change that’s why it is also referred to as a constant-temperature process.
In the Isothermal process, $\text{Change in Temperature} = \Delta T = 0$ or, $T{\text{ }} = \text{constant}$. Graphically, an isothermal process can be represented as: -
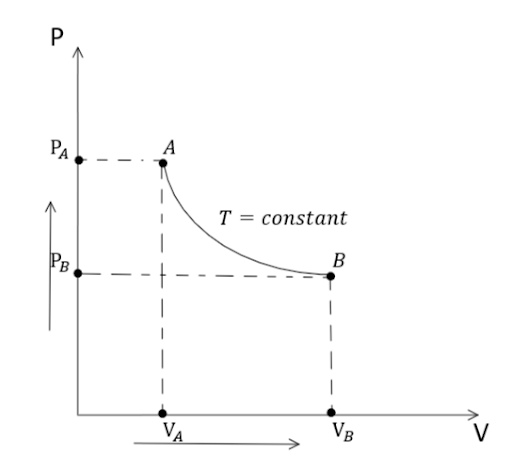
A thermodynamic process is known as an isothermal process when the system temperature is constant because heat is transferred into or out of the system so slowly, that thermal equilibrium is preserved.
If a system comes into contact with a thermal reservoir from the outside, it gradually modifies its own temperature through heat exchange to preserve thermal equilibrium which means heat content is not constant as a result of which options (A) and (B) become incorrect. Thus, in an isothermal process, the temperature remains constant for an ideal gas.
Hence, the correct option is C.
Note: In this case, we know that temperature varies with the given conditions of the system and surroundings. But to keep the system’s temperature constant, either the heat is drawn into the system from the surroundings or it is taken from the system and discharged outside to the surroundings. Additionally, the heat pump is one of the illustrations of an isothermal process.
Complete step by step solution:
The term "isothermal process" refers to a substance, an item, or a system changing at a specific constant temperature. Isothermal Process in thermodynamics is a process during which the temperature $T$ of a system remains constant whereas other variables like pressure $P$ and volume $V$ may change that’s why it is also referred to as a constant-temperature process.
In the Isothermal process, $\text{Change in Temperature} = \Delta T = 0$ or, $T{\text{ }} = \text{constant}$. Graphically, an isothermal process can be represented as: -

A thermodynamic process is known as an isothermal process when the system temperature is constant because heat is transferred into or out of the system so slowly, that thermal equilibrium is preserved.
If a system comes into contact with a thermal reservoir from the outside, it gradually modifies its own temperature through heat exchange to preserve thermal equilibrium which means heat content is not constant as a result of which options (A) and (B) become incorrect. Thus, in an isothermal process, the temperature remains constant for an ideal gas.
Hence, the correct option is C.
Note: In this case, we know that temperature varies with the given conditions of the system and surroundings. But to keep the system’s temperature constant, either the heat is drawn into the system from the surroundings or it is taken from the system and discharged outside to the surroundings. Additionally, the heat pump is one of the illustrations of an isothermal process.
Recently Updated Pages
Mass vs Weight: Key Differences Explained for Students

Uniform Acceleration Explained: Formula, Examples & Graphs

JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 15th Shift 1) Physics Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Physics Chapter 1 - Units And Measurements - 2025-26

NCERT Solutions For Class 11 Physics Chapter 1 Units And Measurements - 2025-26

Important Questions For Class 11 Physics Chapter 1 Units and Measurement - 2025-26

CBSE Notes Class 11 Physics Chapter 4 - Laws of Motion - 2025-26




