When ethyl iodide is heated with silver nitrate, the product obtained is
A. \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{Ag}}\]
B.\[{\rm{Ag}} - {\rm{O}} - {\rm{N}}{{\rm{O}}_{\rm{2}}}\]
C.\[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{O}} - {\rm{N}}{{\rm{O}}_{\rm{2}}}\]
D. \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{I}} - {\rm{N}}{{\rm{O}}_{\rm{2}}}\]
Answer
269.1k+ views
Hint: Silver nitrate is an important organic compound used as an antiseptic and in many chemical reactions. The chemical symbol of silver nitrate is \[{\rm{AgN}}{{\rm{O}}_{\rm{3}}}\] . It is also used in the industrial preparation of many salts of silver.
Complete Step by Step Solution:
Let’s understand the reaction of ethyl iodide and silver nitrate. In the first step, dissociation of bonds of C-I and Ag-N occurs.
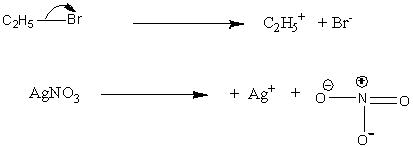
Image: Reaction of ethyl iodide and silver nitrate
In the second step, the nitrate ion attacks the carbocation and the product forms.
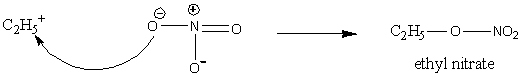
Image: Nucleophilic attack of nitrate ion
Therefore, the product formed in the reaction of ethyl iodide and silver nitrate is ethyl nitrate, \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{O}} - {\rm{N}}{{\rm{O}}_{\rm{2}}}\].
Hence, the option C is right
Additional Information: Let’s discuss ethyl iodide in detail. Ethyl iodide is also chemically known as iodoethane. It is a chemical compound of no colour and flammable properties. The chemical symbol of ethyl iodide is \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{I}}\]. The preparation of ethyl iodide is done by heating ethanol with phosphorus and iodine. The decomposition of iodoethane or ethyl iodide occurs when it comes in contact with air or light.
Note: There are many uses for silver nitrate in our everyday life. Silver nitrate is useful in treating wounds. It is used to treat bone ulcers, burns, and acute wounds. In the laboratory, Silver nitrate is a useful reagent because of its role in the identification of halogens, such as bromine, chlorine etc.
Complete Step by Step Solution:
Let’s understand the reaction of ethyl iodide and silver nitrate. In the first step, dissociation of bonds of C-I and Ag-N occurs.

Image: Reaction of ethyl iodide and silver nitrate
In the second step, the nitrate ion attacks the carbocation and the product forms.
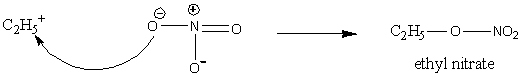
Image: Nucleophilic attack of nitrate ion
Therefore, the product formed in the reaction of ethyl iodide and silver nitrate is ethyl nitrate, \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{O}} - {\rm{N}}{{\rm{O}}_{\rm{2}}}\].
Hence, the option C is right
Additional Information: Let’s discuss ethyl iodide in detail. Ethyl iodide is also chemically known as iodoethane. It is a chemical compound of no colour and flammable properties. The chemical symbol of ethyl iodide is \[{{\rm{C}}_{\rm{2}}}{{\rm{H}}_{\rm{5}}}{\rm{I}}\]. The preparation of ethyl iodide is done by heating ethanol with phosphorus and iodine. The decomposition of iodoethane or ethyl iodide occurs when it comes in contact with air or light.
Note: There are many uses for silver nitrate in our everyday life. Silver nitrate is useful in treating wounds. It is used to treat bone ulcers, burns, and acute wounds. In the laboratory, Silver nitrate is a useful reagent because of its role in the identification of halogens, such as bromine, chlorine etc.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




