Each of the following options contains a set of four molecules, identify the option(s) where all four molecules possess permanent dipole moment at room temperature.
This question has multiple correct options
(A) $N{{O}_{2,}}N{{H}_{3}},POC{{l}_{3}},C{{H}_{3}}Cl$
(B) $B{{F}_{3}},{{O}_{3}},S{{F}_{6}},Xe{{F}_{6}}$
(C) $BeC{{l}_{2}},C{{O}_{2}},BC{{l}_{3}}_{,}CHC{{l}_{3}}$
(D) $S{{O}_{2}},{{C}_{6}}{{H}_{5}}Cl,{{H}_{2}}Se,Br{{F}_{5}}$
Answer
249.6k+ views
Hint: Dipole moments occur when there is a separation of charge and it is a measure of the polarity of the molecule. Among the given molecules $BeC{{l}_{2}},C{{O}_{2}},BC{{l}_{3}},S{{F}_{6}}$and $B{{F}_{3}}$ are non-polar which means that they do not have permanent dipole moment.
Complete step by step solution:
- As we know, polar character is the separation of negative and positive charges in a compound and the measurement of this polar character of a chemical bond in a molecule or between two atoms is given by dipole moment.
- In any system the separation of charges will lead to the formation of dipole moment and the main reason for the development of dipole moment is the differences in electronegativity between the chemically bonded atoms.
- Although the net charge on a molecule is zero, the nature of chemical bonds is such that the negative and positive charges do not totally overlap in most molecules and such molecules are said to be polar because they possess a permanent dipole moment($\mu $).
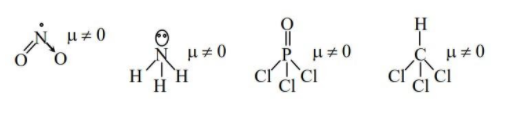
- As we can see all the molecules have nonzero dipole moment and hence they possess permanent dipole moment. Hence option (A) is correct.
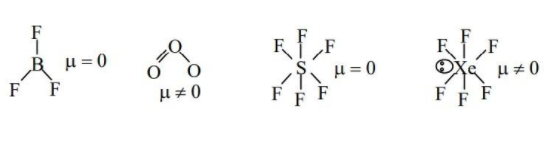
- The net dipole moment of the molecules$B{{F}_{3}}$ and $S{{F}_{6}}$ is zero and hence they do not possess permanent dipole moments. Hence option (B) is incorrect.
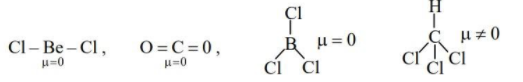
-Only the molecule $CHC{{l}_{3}}$ has permanent dipole moment. Hence option (C) is also incorrect.
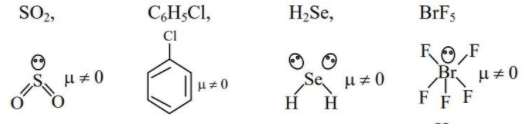
As we can see all the molecules have nonzero dipole moment and hence they possess permanent dipole moment. Hence option (D) is correct.
Thus the set of molecules $N{{O}_{2,}}N{{H}_{3}},POC{{l}_{3}},C{{H}_{3}}Cl$ and $S{{O}_{2}},{{C}_{6}}{{H}_{5}}Cl,{{H}_{2}}Se,Br{{F}_{5}}$ possess permanent dipole moment at room temperature.
Therefore the answers are options (A) and (D).
Note: The difference between the terms bond dipole moment and total dipole moment should be noted. The total dipole moment in a polyatomic molecule is the vector sum of all the bond dipoles whereas the bond dipole moment is the dipole moment between the single bonds of a diatomic molecule.
Complete step by step solution:
- As we know, polar character is the separation of negative and positive charges in a compound and the measurement of this polar character of a chemical bond in a molecule or between two atoms is given by dipole moment.
- In any system the separation of charges will lead to the formation of dipole moment and the main reason for the development of dipole moment is the differences in electronegativity between the chemically bonded atoms.
- Although the net charge on a molecule is zero, the nature of chemical bonds is such that the negative and positive charges do not totally overlap in most molecules and such molecules are said to be polar because they possess a permanent dipole moment($\mu $).

- As we can see all the molecules have nonzero dipole moment and hence they possess permanent dipole moment. Hence option (A) is correct.

- The net dipole moment of the molecules$B{{F}_{3}}$ and $S{{F}_{6}}$ is zero and hence they do not possess permanent dipole moments. Hence option (B) is incorrect.

-Only the molecule $CHC{{l}_{3}}$ has permanent dipole moment. Hence option (C) is also incorrect.

As we can see all the molecules have nonzero dipole moment and hence they possess permanent dipole moment. Hence option (D) is correct.
Thus the set of molecules $N{{O}_{2,}}N{{H}_{3}},POC{{l}_{3}},C{{H}_{3}}Cl$ and $S{{O}_{2}},{{C}_{6}}{{H}_{5}}Cl,{{H}_{2}}Se,Br{{F}_{5}}$ possess permanent dipole moment at room temperature.
Therefore the answers are options (A) and (D).
Note: The difference between the terms bond dipole moment and total dipole moment should be noted. The total dipole moment in a polyatomic molecule is the vector sum of all the bond dipoles whereas the bond dipole moment is the dipole moment between the single bonds of a diatomic molecule.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




