Draw the Lewis structure for \[IC{l_4}^ - \] and provide the following information.
A. Total number of valence electrons
B. Number of nonbonding electron pairs
C. Electron geometry
D. Molecular geometry
E. Polarity
Answer
264k+ views
Hint: VSEPR theory is used to understand the arrangement of electrons. Here the focus is on the arrangements around Iodine in \[IC{l_4}^ - \]. Once the molecular geometry is understood, the Polarity of molecules/species can be calculated.
Lewis’s structure will enable us to determine a stable bonding arrangement for this ion.
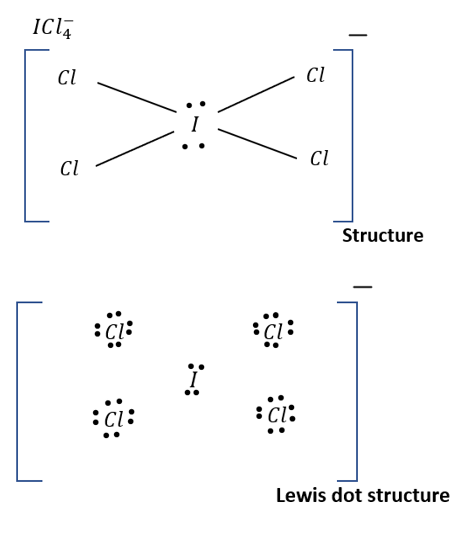
Complete step by step solution:
(a) Total number of valence electrons.
Iodine has 7 valence electrons and chlorine has also 7 electrons in its outermost orbit. One more negative charge on the outside. Hence, total number of valence electrons is:
\[V_e = 7 + 7[4] + 1\\ = 36\]
On the periodic chart, iodine [I] is in Period 3 and may hold more than 8 electrons. The Iodine atom contains 12 valence electrons in the Lewis structure for ICl4-.
(b) Number of nonbonding electron pairs:
As per Lewis structure, chlorine has 6 electrons which does not form bond and iodine has 4 electrons which do not form bond
Therefore, there are 2 non-bonding \[\bar e\] pairs on Iodine & \[3\] non-bonding \[\bar e\] pairs on each chlorine.
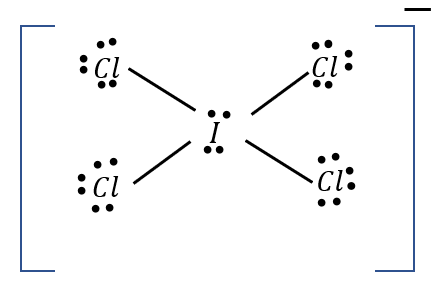
Image: Lewis structure of \[IC{l_4}^ - \]
A total of \[14\] non-bonding \[\bar e\] pairs are present in this .
(c) Electron geometry
It has octahedral electron geometry.
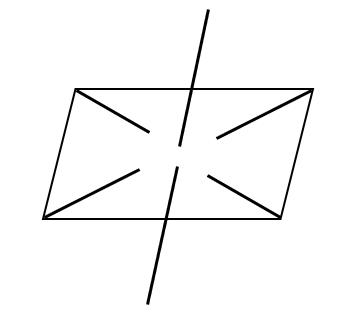
Image: Electron geometry of \[IC{l_4}^ - \]
(d) Molecular geometry
It has square planar geometry, because \[2\]non-bonding \[\bar e\] pairs of Iodine are occupying \[2\]opposite vertices.
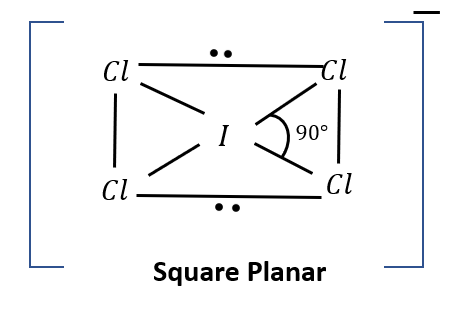
Image: Molecular geometry of \[IC{l_4}^ - \]
[e] Polarity:
It is a non-polar compound because it is a square plane. In \[ICl\] bond, \[Cl\] is the most negative, so it attracts the electrons into chemical bonds.
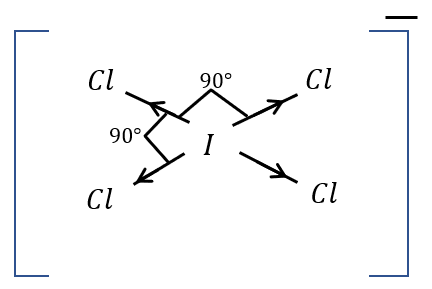
Image: Polarity of \[IC{l_4}^ - \]
There is polarity in each \[I - Cl\] bond. But overall polarity is cancelled out due to geometry.
Note: Iron reacting with hydrochloric acid forms iron chloride where iron is in +2 oxidation state whereas when iron reacts with chlorine it also forms iron chloride but here iron is in +3oxidation state. The reaction between iron and chlorine is a type of redox reaction. The reaction between iron and chlorine is shown below:
$2Fe(s) + 3C{l_2}(g) \to 2FeC{l_3}(s)$
Lewis’s structure will enable us to determine a stable bonding arrangement for this ion.

Complete step by step solution:
(a) Total number of valence electrons.
Iodine has 7 valence electrons and chlorine has also 7 electrons in its outermost orbit. One more negative charge on the outside. Hence, total number of valence electrons is:
\[V_e = 7 + 7[4] + 1\\ = 36\]
On the periodic chart, iodine [I] is in Period 3 and may hold more than 8 electrons. The Iodine atom contains 12 valence electrons in the Lewis structure for ICl4-.
(b) Number of nonbonding electron pairs:
As per Lewis structure, chlorine has 6 electrons which does not form bond and iodine has 4 electrons which do not form bond
Therefore, there are 2 non-bonding \[\bar e\] pairs on Iodine & \[3\] non-bonding \[\bar e\] pairs on each chlorine.

Image: Lewis structure of \[IC{l_4}^ - \]
A total of \[14\] non-bonding \[\bar e\] pairs are present in this .
(c) Electron geometry
It has octahedral electron geometry.

Image: Electron geometry of \[IC{l_4}^ - \]
(d) Molecular geometry
It has square planar geometry, because \[2\]non-bonding \[\bar e\] pairs of Iodine are occupying \[2\]opposite vertices.

Image: Molecular geometry of \[IC{l_4}^ - \]
[e] Polarity:
It is a non-polar compound because it is a square plane. In \[ICl\] bond, \[Cl\] is the most negative, so it attracts the electrons into chemical bonds.

Image: Polarity of \[IC{l_4}^ - \]
There is polarity in each \[I - Cl\] bond. But overall polarity is cancelled out due to geometry.
Note: Iron reacting with hydrochloric acid forms iron chloride where iron is in +2 oxidation state whereas when iron reacts with chlorine it also forms iron chloride but here iron is in +3oxidation state. The reaction between iron and chlorine is a type of redox reaction. The reaction between iron and chlorine is shown below:
$2Fe(s) + 3C{l_2}(g) \to 2FeC{l_3}(s)$
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




