Why does \[N{H_3}\] act as a Lewis base?
Answer
264.3k+ views
Hint : A Lewis base is a substance that can donate a pair of non bonding electrons. A Lewis base is therefore called electron-pair-donor. In Lewis base molecules the orbitals used to be highly occupied so they have a tendency to donate electrons. The most common Lewis base is ammonia .
Complete answer:
> In ammonia the molecule consists of a nitrogen atom as a central metal atom. Ammonia is Lewis base because nitrogen has a lone pair of electrons which can be donated, hence it acts as Lewis base. \[N{H_3}\] can give a lone pair of electrons to protons to form ammonium ions . This reaction shows its Lewis base tendency. $N{H_3} + {H^ + } \to N{H^ + }_4$ $ \Rightarrow $ Here in this reaction the lone pair on the nitrogen atom is transferred to the hydrogen ion . Hydronium ion acts as a Lewis acid here which accepts electrons from ammonia.
> A Lewis acid-base reaction can be done when a Lewis base donates a pair of electrons to an Lewis acid. Lets see an example of Lewis acid-base reaction. In the following reaction each of two ammonia molecules which is a Lewis base donates a pair of electrons to a silver ion which acts as Lewis acid in the reaction.
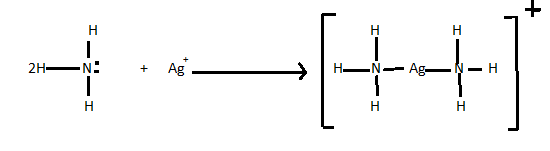 There are so many reactions that are Lewis acid-base displacement reactions.
There are so many reactions that are Lewis acid-base displacement reactions.
Note : Here we learned about Lewis base and also learned why ammonia acts as a Lewis base in any reaction. Ammonia has a filled orbital containing an electron pair which does not involve bonding and Lewis acid contains an empty orbital which is capable of accepting electrons.
Complete answer:
> In ammonia the molecule consists of a nitrogen atom as a central metal atom. Ammonia is Lewis base because nitrogen has a lone pair of electrons which can be donated, hence it acts as Lewis base. \[N{H_3}\] can give a lone pair of electrons to protons to form ammonium ions . This reaction shows its Lewis base tendency. $N{H_3} + {H^ + } \to N{H^ + }_4$ $ \Rightarrow $ Here in this reaction the lone pair on the nitrogen atom is transferred to the hydrogen ion . Hydronium ion acts as a Lewis acid here which accepts electrons from ammonia.
> A Lewis acid-base reaction can be done when a Lewis base donates a pair of electrons to an Lewis acid. Lets see an example of Lewis acid-base reaction. In the following reaction each of two ammonia molecules which is a Lewis base donates a pair of electrons to a silver ion which acts as Lewis acid in the reaction.
 There are so many reactions that are Lewis acid-base displacement reactions.
There are so many reactions that are Lewis acid-base displacement reactions.Note : Here we learned about Lewis base and also learned why ammonia acts as a Lewis base in any reaction. Ammonia has a filled orbital containing an electron pair which does not involve bonding and Lewis acid contains an empty orbital which is capable of accepting electrons.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




