\[Diazonium~salts + C{u_2}C{l_2} + HCl \to \]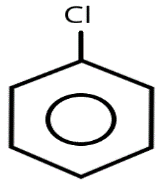 , the reaction is known as
, the reaction is known as
A. Chlorination
B. Sandmeyer’s reaction
C. Perkin reaction
D. Carbyl amine reaction
Answer
269.4k+ views
Hint: A group of organic compounds known as diazonium salts have a functional group \[[RN_2]X\] that can have any kind of organic group \[R\], like an aryl or an alkyl, and an inorganic or organic anion, like a halide. It is used in many multiple reactions to form new organic compounds.
Complete step-by-step answer:Diazotization is a chemical reaction in which amine gets converted into diazonium salt with the help of sodium nitrite and HCl at low temperatures of 0-5 degree celsius. The creation of the azo compound is caused by the reaction of benzene-diazonium chloride with another chemical containing a benzene ring known as a coupling specialist, such as phenol or aromatic amine.
By heating aryl azide and cuprous halide, Sandmeyer's reaction produces halobenzene.
Halobenzene is created using Sandmeyer's reaction. For the creation of alkyl fluorides, it cannot be employed. It is an illustration of an aromatic radical-nucleophilic substitution. The Sandmeyer’s reaction for the formation of chlorobenzene is shown below.
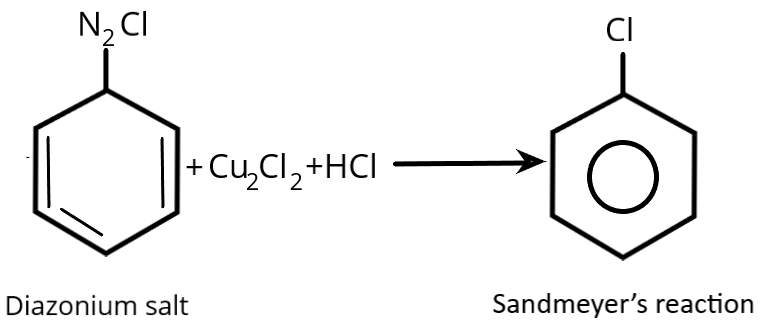
Therefore, the given reaction represents Sandmeyer's reaction.
Option ‘B’ is correct
Note:It should be noted that the finding of biaryl byproducts lends support to the radical mechanism of the Sandmeyer reaction. A one-electron transfer mechanism that is catalysed by copper\[(I)\] causes the aromatic diazo group to be replaced with a halogen or pseudohalogen, forming an aryl radical with the loss of nitrogen gas. The substitutes may be produced and the copper\[(I)\] catalyst restored by direct transfer of \[Cl,Br\] or \[OH\] from a copper\[(II)\] species to the aryl radical.
Complete step-by-step answer:Diazotization is a chemical reaction in which amine gets converted into diazonium salt with the help of sodium nitrite and HCl at low temperatures of 0-5 degree celsius. The creation of the azo compound is caused by the reaction of benzene-diazonium chloride with another chemical containing a benzene ring known as a coupling specialist, such as phenol or aromatic amine.
By heating aryl azide and cuprous halide, Sandmeyer's reaction produces halobenzene.
Halobenzene is created using Sandmeyer's reaction. For the creation of alkyl fluorides, it cannot be employed. It is an illustration of an aromatic radical-nucleophilic substitution. The Sandmeyer’s reaction for the formation of chlorobenzene is shown below.

Therefore, the given reaction represents Sandmeyer's reaction.
Option ‘B’ is correct
Note:It should be noted that the finding of biaryl byproducts lends support to the radical mechanism of the Sandmeyer reaction. A one-electron transfer mechanism that is catalysed by copper\[(I)\] causes the aromatic diazo group to be replaced with a halogen or pseudohalogen, forming an aryl radical with the loss of nitrogen gas. The substitutes may be produced and the copper\[(I)\] catalyst restored by direct transfer of \[Cl,Br\] or \[OH\] from a copper\[(II)\] species to the aryl radical.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Understanding Atomic Structure for Beginners

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




