DDT can be prepared by reacting chlorobenzene in the presence of conc.\[{H_2}S{O_4}\] with ______.
Answer
265.5k+ views
Hint: The full form of DDT is dichlorodiphenyltrichloroethane. DDT is a colourless, odourless, tasteless, crystalline chemical compound. The compound to which it reacts is an aldehyde.
Complete Step by Step Solution:
DDT or dichlorodiphenyltrichloroethane is used as an insecticide in agriculture. The chemical formula of DDT is \[{C_{14}}{H_9}C{l_5}\] . The compound is present as a colourless and tasteless crystalline solid in its standard condition for temperature and pressure.
DDT is prepared by reacting chloral and chlorobenzene in presence of concentrated sulphuric acid in a ratio of 1:2.
Chloral is an organic compound having a molecular formula of \[C{l_3}CCHO\]. The other name of chloral is trichloroacetaldehyde or trichloroethanal. It is a colourless oily aldehyde compound which shows solubility in most solvents.
The reaction between chlorobenzene and chloral is shown below.
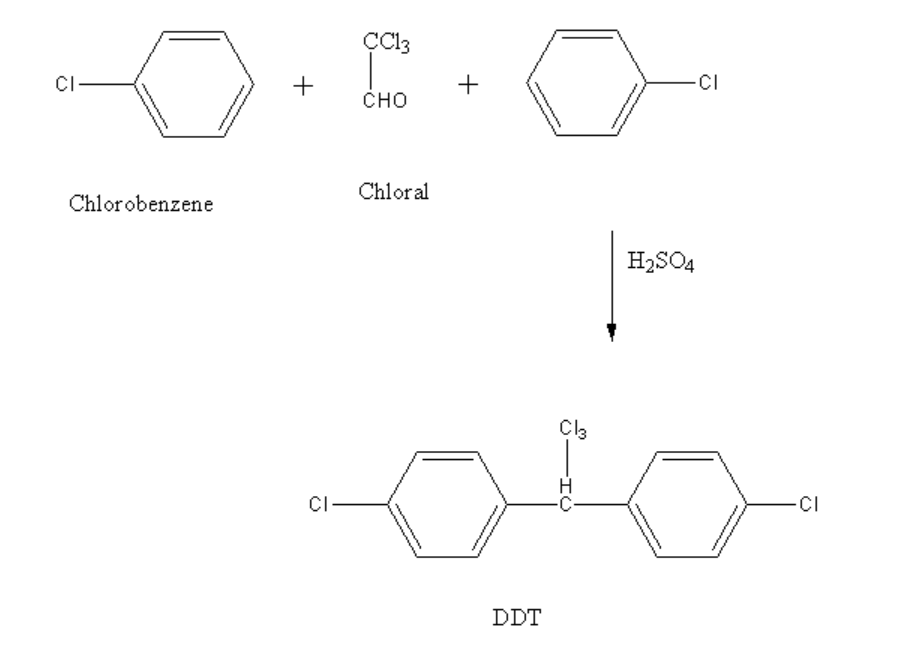
Image: Preparation of DDT
In the above reaction, two moles of chlorobenzene reacts with one mole of chloral in presence of concentrated sulphuric acid to form dichlorodiphenyltrichloroethane.
Therefore, DDT can be prepared by reacting chlorobenzene in the presence of conc.\[{H_2}S{O_4}\] with chloral.
Additional information: DDT is now banned in different countries as it is a very toxic compound and due to its high usage it leads to death. Chloral on dissolving in water forms chloral hydrate.
Note: Sometimes it may be confusing while drawing the structure of DDT as it is a germinal compound where two benzyl chloride group is attached to one carbon and only one chlorine group is on the other carbon atom.
Complete Step by Step Solution:
DDT or dichlorodiphenyltrichloroethane is used as an insecticide in agriculture. The chemical formula of DDT is \[{C_{14}}{H_9}C{l_5}\] . The compound is present as a colourless and tasteless crystalline solid in its standard condition for temperature and pressure.
DDT is prepared by reacting chloral and chlorobenzene in presence of concentrated sulphuric acid in a ratio of 1:2.
Chloral is an organic compound having a molecular formula of \[C{l_3}CCHO\]. The other name of chloral is trichloroacetaldehyde or trichloroethanal. It is a colourless oily aldehyde compound which shows solubility in most solvents.
The reaction between chlorobenzene and chloral is shown below.

Image: Preparation of DDT
In the above reaction, two moles of chlorobenzene reacts with one mole of chloral in presence of concentrated sulphuric acid to form dichlorodiphenyltrichloroethane.
Therefore, DDT can be prepared by reacting chlorobenzene in the presence of conc.\[{H_2}S{O_4}\] with chloral.
Additional information: DDT is now banned in different countries as it is a very toxic compound and due to its high usage it leads to death. Chloral on dissolving in water forms chloral hydrate.
Note: Sometimes it may be confusing while drawing the structure of DDT as it is a germinal compound where two benzyl chloride group is attached to one carbon and only one chlorine group is on the other carbon atom.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




