Cyclopentene on treatment with alkaline potassium permanganate gives
A. Cyclopentanol
B. Trans 1, 2-cyclopentanediol
C. Cis 1, 2-cyclopentanediol
D. 1:1 mixture of cis and Trans-1,2-cyclopentanediol
Answer
266.4k+ views
Hint: Cycloalkenes are ring compounds with one or more double bonds in the molecule. Cyclopentene is a type of cycloalkenes. Cyclopentene on treatment with alkaline potassium permanganate undergoes oxidation as alkaline potassium permanganate is a strong oxidising agent.
Complete Step by Step Answer:
Cycloalkenes are cyclic compounds possessing one or more double bonds in the molecule. Cyclopentene is a type of cycloalkenes.
Potassium manganate(VII) solution when added to sodium carbonate solution becomes little alkaline. The purple solution initially becomes dark green and then develops a dark brown precipitate. Alkaline potassium permanganate is a strong oxidising agent which undergoes reduction itself.
Under an alkaline climate, the manganate (VII) ions are initially reduced to green manganate (VI) ions.
Manganate(VII) ions are a powerful oxidising agent and oxidise cyclopentene to cis 1,2-cyclopentanediol.
The alkaline potassium manganate (VII) solution makes the cyclopentene undergo oxidation by breaking down the carbon-carbon double bond and altering it with two bonds in which carbon is attached to the hydroxyl group. This is a cis isomer because two bonds in which carbon is attached to the hydroxyl group
bonds are on the same side.
The reaction happens as follows:-
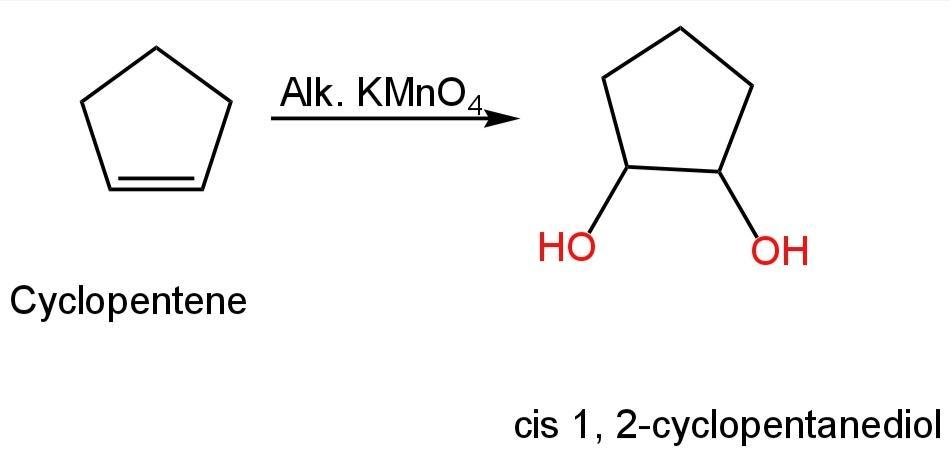
Image: Conversion of cyclopentene into cis 1, 2-cyclopentanediol.
So, option C is correct.
Note: Alkenes react with potassium manganate(VII) solution. It is a purple colour solution. The colour modification relies on whether the potassium manganate(VII) is utilised in acidic or alkaline conditions.If the potassium manganate(VII) solution is mixed with dilute sulfuric acid, the purple solution becomes colourless.
Complete Step by Step Answer:
Cycloalkenes are cyclic compounds possessing one or more double bonds in the molecule. Cyclopentene is a type of cycloalkenes.
Potassium manganate(VII) solution when added to sodium carbonate solution becomes little alkaline. The purple solution initially becomes dark green and then develops a dark brown precipitate. Alkaline potassium permanganate is a strong oxidising agent which undergoes reduction itself.
Under an alkaline climate, the manganate (VII) ions are initially reduced to green manganate (VI) ions.
Manganate(VII) ions are a powerful oxidising agent and oxidise cyclopentene to cis 1,2-cyclopentanediol.
The alkaline potassium manganate (VII) solution makes the cyclopentene undergo oxidation by breaking down the carbon-carbon double bond and altering it with two bonds in which carbon is attached to the hydroxyl group. This is a cis isomer because two bonds in which carbon is attached to the hydroxyl group
bonds are on the same side.
The reaction happens as follows:-

Image: Conversion of cyclopentene into cis 1, 2-cyclopentanediol.
So, option C is correct.
Note: Alkenes react with potassium manganate(VII) solution. It is a purple colour solution. The colour modification relies on whether the potassium manganate(VII) is utilised in acidic or alkaline conditions.If the potassium manganate(VII) solution is mixed with dilute sulfuric acid, the purple solution becomes colourless.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




