Cyclohexene on reaction with OsO4 followed by reaction with NaHSO3 gives
A. Cis-diol
B. Trans-diol
C. Epoxy
D. Alcohol
Answer
268.2k+ views
Hint: The process of conversion of alkene into vicinal diol is called dihydroxylation. It is an oxidation process and for this process, a transition metal with a high oxidation state is needed like osmium or manganese. OsO4 is the most important oxidising agent which is used for the process of dihydroxylation.
Complete Step by Step Answer:
The reaction of cyclohexane with OsO4 is a dihydroxylation reaction, dihydroxylation means the addition of two hydroxyl groups in the substituent, and the formation of vicinal diol takes place.
The reaction of Cyclohexene with OsO4 followed by a reaction with NaHSO3 will give a diol-
$C_6H_{10}\xrightarrow[]{OsO_4/NaHSO_3}C_6H_{10}\left ( OH \right )_2$
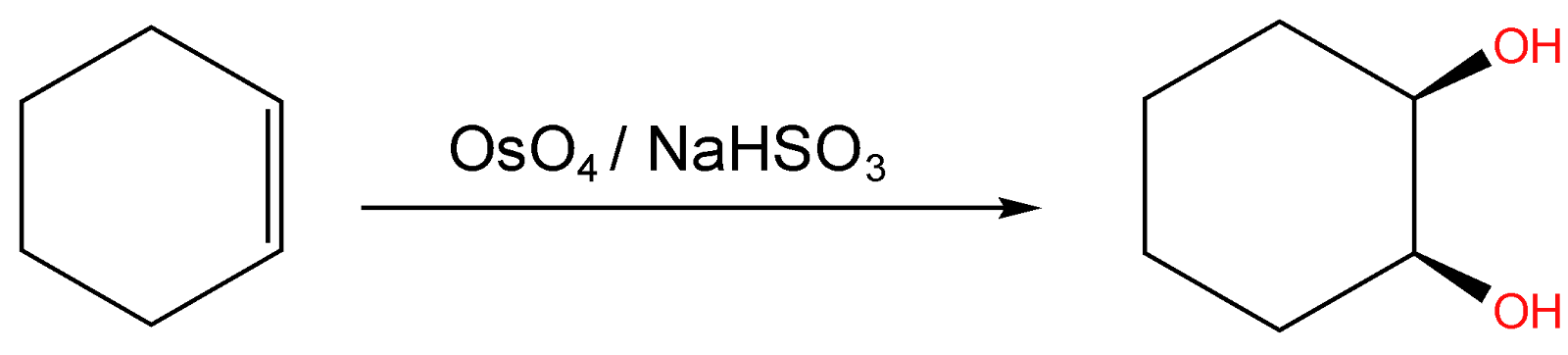
This is an oxidative addition reaction. And the mechanism of the reaction passes through the transition state and the formation of the transition state requires syn addition thus the product formed will be syn.
The reaction mechanism is
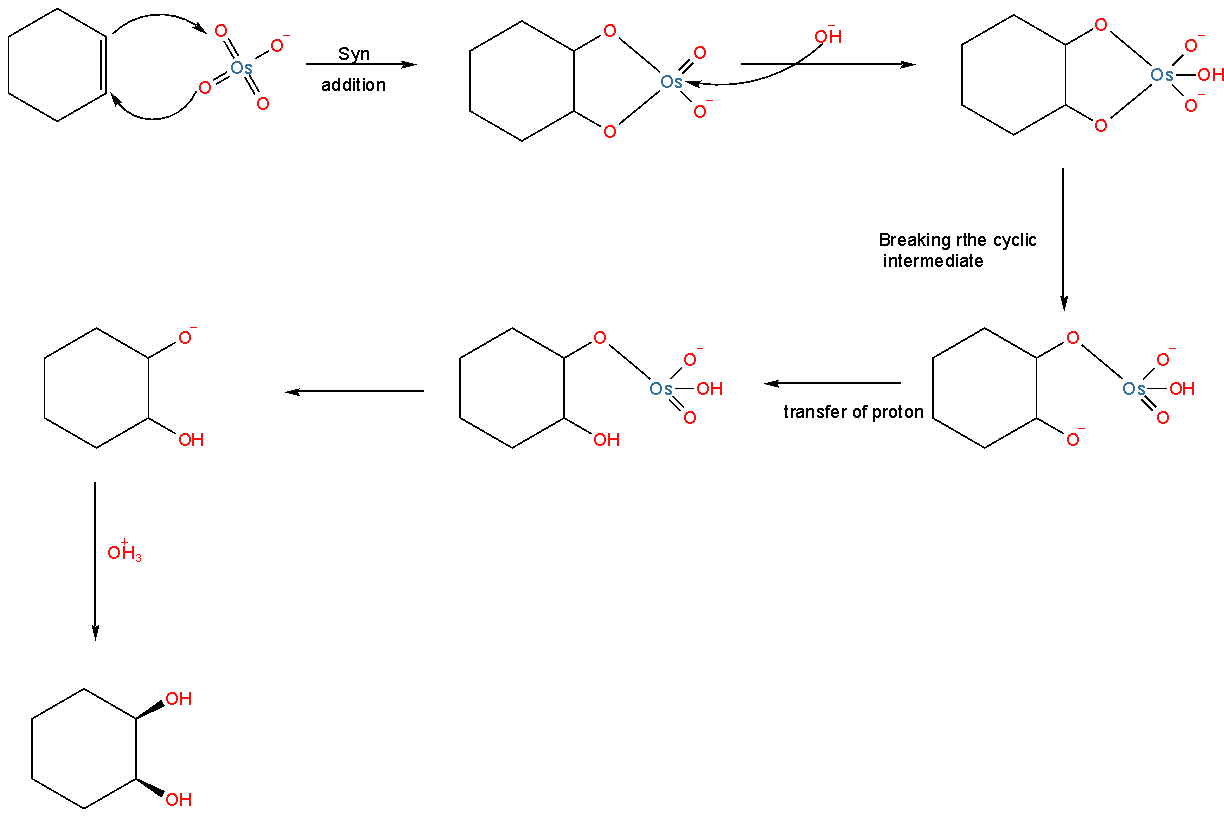
This process follows no rearrangement. The reaction follows syn addition, that is the addition of a new bond will be at the same face of the reactant. In this reaction, NaHSO3 is used to break the five-membered cyclic transition state.
Thus we can see that the Cyclohexene on reaction with OsO4 followed by reaction with NaHSO3 gives cyclohexane cis diol.
Thus, Option (A) is correct
Note: Bayer’s reagent which is alkaline potassium permanganate (alkaline) KMnO4 is also a strong oxidising agent which is also used for the formation of vicinal diol. Manganese also has a high oxidation state. It is also used for the determination of unsaturation in a compound.
Complete Step by Step Answer:
The reaction of cyclohexane with OsO4 is a dihydroxylation reaction, dihydroxylation means the addition of two hydroxyl groups in the substituent, and the formation of vicinal diol takes place.
The reaction of Cyclohexene with OsO4 followed by a reaction with NaHSO3 will give a diol-
$C_6H_{10}\xrightarrow[]{OsO_4/NaHSO_3}C_6H_{10}\left ( OH \right )_2$

This is an oxidative addition reaction. And the mechanism of the reaction passes through the transition state and the formation of the transition state requires syn addition thus the product formed will be syn.
The reaction mechanism is

This process follows no rearrangement. The reaction follows syn addition, that is the addition of a new bond will be at the same face of the reactant. In this reaction, NaHSO3 is used to break the five-membered cyclic transition state.
Thus we can see that the Cyclohexene on reaction with OsO4 followed by reaction with NaHSO3 gives cyclohexane cis diol.
Thus, Option (A) is correct
Note: Bayer’s reagent which is alkaline potassium permanganate (alkaline) KMnO4 is also a strong oxidising agent which is also used for the formation of vicinal diol. Manganese also has a high oxidation state. It is also used for the determination of unsaturation in a compound.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




