Cyclohexane, methylcyclopentane, 1, 3-dimethyl cyclobutane and 1, 2, 3- trimethyl cyclopropane are examples of which one of the following?
A. Constitutional isomers
B. Positional isomers
C. Structural isomers
D. Structural as well as positional isomers
Answer
260.4k+ views
Hint: We know that isomers are different in physical and chemical properties but have the same number of atoms. This phenomenon is termed as isomerism.
Complete step by step answer:
Structural isomers are those isomers where atoms are fully arranged with the same molecular formulae in different order. Molecular formula of Cyclohexane, methylcyclopentane, 1, 3-dimethyl cyclobutane and 1, 2, 3- trimethyl cyclopropane is ${{\rm{C}}_{\rm{6}}}{{\rm{H}}_{{\rm{12}}}}$.
Cyclohexane has 6 carbon and 12 hydrogen atoms. Now we draw structure of cyclohexane is as follows:
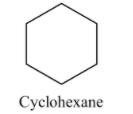
Methylcyclopentane has 6 carbon and 12 hydrogen atoms. Now we draw structure of methylcyclopentane is as follows:
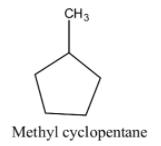
1, 3-dimethyl cyclobutane has 6 carbon and 12 hydrogen atoms. Now we draw structure of 1, 3-dimethyl cyclobutane is as follows:
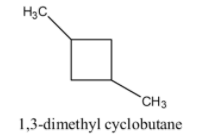
1, 2, 3- trimethyl cyclopropane has 6 carbon and 12 hydrogen atoms. Now we draw structure of 1, 2, 3- trimethyl cyclopropane is as follows:
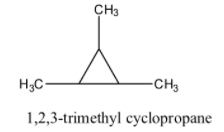
Structural isomers are also termed as constitutional isomers. Constitutional isomers have the same molecular formula but numbering and IUPAC names are different. Only count the number of each atom in both molecules to see how atoms are organized to decide whether two molecules are constitutional isomers.
Position isomers are also structural or constitutional isomers with the same functional group and same carbon skeleton but differing in position of the same functional group on or inside the carbon chain.
Cyclohexane, methylcyclopentane, 1, 3-dimethyl cyclobutane and 1, 2, 3- trimethyl cyclopropane are structural isomers because they have same molecular formula $\left( {{{\rm{C}}_{\rm{6}}}{{\rm{H}}_{{\rm{12}}}}} \right)$ but different bonding arrangement.
Hence, the correct answer is C.
Note:
Isomers are classified as structural (constitutional) and stereoisomerism. Structural isomers are further classified as chain isomers, position isomers and functional group isomers.
Complete step by step answer:
Structural isomers are those isomers where atoms are fully arranged with the same molecular formulae in different order. Molecular formula of Cyclohexane, methylcyclopentane, 1, 3-dimethyl cyclobutane and 1, 2, 3- trimethyl cyclopropane is ${{\rm{C}}_{\rm{6}}}{{\rm{H}}_{{\rm{12}}}}$.
Cyclohexane has 6 carbon and 12 hydrogen atoms. Now we draw structure of cyclohexane is as follows:

Methylcyclopentane has 6 carbon and 12 hydrogen atoms. Now we draw structure of methylcyclopentane is as follows:

1, 3-dimethyl cyclobutane has 6 carbon and 12 hydrogen atoms. Now we draw structure of 1, 3-dimethyl cyclobutane is as follows:

1, 2, 3- trimethyl cyclopropane has 6 carbon and 12 hydrogen atoms. Now we draw structure of 1, 2, 3- trimethyl cyclopropane is as follows:

Structural isomers are also termed as constitutional isomers. Constitutional isomers have the same molecular formula but numbering and IUPAC names are different. Only count the number of each atom in both molecules to see how atoms are organized to decide whether two molecules are constitutional isomers.
Position isomers are also structural or constitutional isomers with the same functional group and same carbon skeleton but differing in position of the same functional group on or inside the carbon chain.
Cyclohexane, methylcyclopentane, 1, 3-dimethyl cyclobutane and 1, 2, 3- trimethyl cyclopropane are structural isomers because they have same molecular formula $\left( {{{\rm{C}}_{\rm{6}}}{{\rm{H}}_{{\rm{12}}}}} \right)$ but different bonding arrangement.
Hence, the correct answer is C.
Note:
Isomers are classified as structural (constitutional) and stereoisomerism. Structural isomers are further classified as chain isomers, position isomers and functional group isomers.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




