Conversion of phenol to salicylic acid and to salicyaldehyde are known as? (respectively)
(A) Reimer-Tiemann reaction and Kolbe’s reaction
(B) Williamson’s synthesis and Hydroboration-oxidation
(C) Kolbe’s reaction and Williamson’s synthesis
(D) Kolbe’s reaction and Reimer-Tiemann reaction
Answer
560.8k+ views
Hint:The conversion of phenol to salicylic acid takes place through a base, and hydrolysis. If we talk about the conversion of phenol of salicylaldehyde takes place through the action of chloroform. So, we identify the naming reactions in which these conversions take place.
Complete step by step solution:
First, we will discuss the conversion of phenol to salicylic acid.
The phenol will be deprotonated by strong base i.e. hydroxide ion. There will be addition of phenoxide ions formed to the carbon-dioxide.
So, we can say that the addition further leads to the carboxylate formation. Then, it will further lead to the formation of carboxylic acid, i.e. salicylic acid.
The chemical reaction is
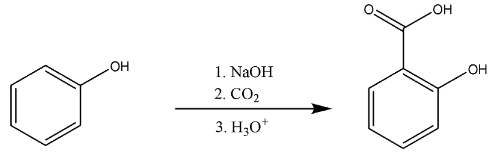
Thus, we know this whole mechanism takes place in Kolbe’s reaction, it is used for the synthesis of aspirin also known as salicylic acid in the industries.
Now, the next we have conversion of phenol to salicylaldehyde. It was proposed by Karl Riemer, mainly used for the ortho- phenols.
In this reaction too like Kolbe’s reaction, there is deprotonation of chloroform by strong base like KOH.
This reaction takes place through the nucleophilic attack, and the basic hydrolysis is done, leading to the formation of salicylaldehyde.
The chemical reaction is
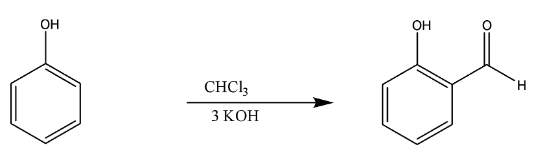
Thus, we can say this whole mechanism takes place in the Riemer –Tiemann reaction.
In the last, we can conclude that conversion of phenol to salicylic acid and to salicylaldehyde are known as Kolbe’ reaction, and Riemer-Tiemann reaction.
Hence, the correct option is (D).
Note: Don’t get confused between both these reactions. These both takes place through deprotonation by a strong base, as we discussed; but the product formation will be different. In case 1 there is addition of carbon-dioxide, and in case 2 there is nucleophilic attack.
Complete step by step solution:
First, we will discuss the conversion of phenol to salicylic acid.
The phenol will be deprotonated by strong base i.e. hydroxide ion. There will be addition of phenoxide ions formed to the carbon-dioxide.
So, we can say that the addition further leads to the carboxylate formation. Then, it will further lead to the formation of carboxylic acid, i.e. salicylic acid.
The chemical reaction is

Thus, we know this whole mechanism takes place in Kolbe’s reaction, it is used for the synthesis of aspirin also known as salicylic acid in the industries.
Now, the next we have conversion of phenol to salicylaldehyde. It was proposed by Karl Riemer, mainly used for the ortho- phenols.
In this reaction too like Kolbe’s reaction, there is deprotonation of chloroform by strong base like KOH.
This reaction takes place through the nucleophilic attack, and the basic hydrolysis is done, leading to the formation of salicylaldehyde.
The chemical reaction is

Thus, we can say this whole mechanism takes place in the Riemer –Tiemann reaction.
In the last, we can conclude that conversion of phenol to salicylic acid and to salicylaldehyde are known as Kolbe’ reaction, and Riemer-Tiemann reaction.
Hence, the correct option is (D).
Note: Don’t get confused between both these reactions. These both takes place through deprotonation by a strong base, as we discussed; but the product formation will be different. In case 1 there is addition of carbon-dioxide, and in case 2 there is nucleophilic attack.
Recently Updated Pages
Area of an Octagon Formula Explained Simply

Absolute Pressure Formula Explained: Key Equation & Examples

Difference Between Atom and Molecule: JEE Main 2026

Difference Between Vapor and Gas: JEE Main 2026

Carbon Dioxide Formula - Definition, Uses and FAQs

Central Angle of a Circle Formula Explained Quickly

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




