Chloroform when treated with aniline and alcoholic KOH gives:
a) phenyl cyanide
b) iso-phenyl cyanide
c) chlorobenzene
d) phenol
Answer
249.9k+ views
Hint The reaction of Chloroform which is treated with aniline and alcoholic KOH. Look that it is the isocyanide test. From the name we will guess the isocyanide is the product because in between the reactants nobody is isocyanide.
Complete step-by-step solution
The reaction of Chloroform with aniline and alcoholic KOH is called isocyanide test because the product is an iso-cyanide. Let’s know something more about the isocyanide test.
Aniline is an aromatic amine. It is a precursor to various drugs, dyes, and plastics. It is also a pollutant. Aniline is toxic, and it can be harmful to the environment.
The isocyanide test can be used to determine the presence of aniline in a sample. The test uses a reagent that reacts with aniline to form a blue colour. The reaction is not specific for aniline, so other compounds that contain aniline-like structures can also produce a blue colour. Only, primary aliphatic and aromatic amines provide it.
The isocyanide test is a quantitative test. The test can be used to determine the concentration of aniline in a sample. The test is also sensitive to low concentrations of aniline.
Now let’s see the product of the reaction of aniline and chloroform with alcoholic KOH. Obviously, the product is an isocyanide that iso-phenyl cyanide. Let's see the reaction now.
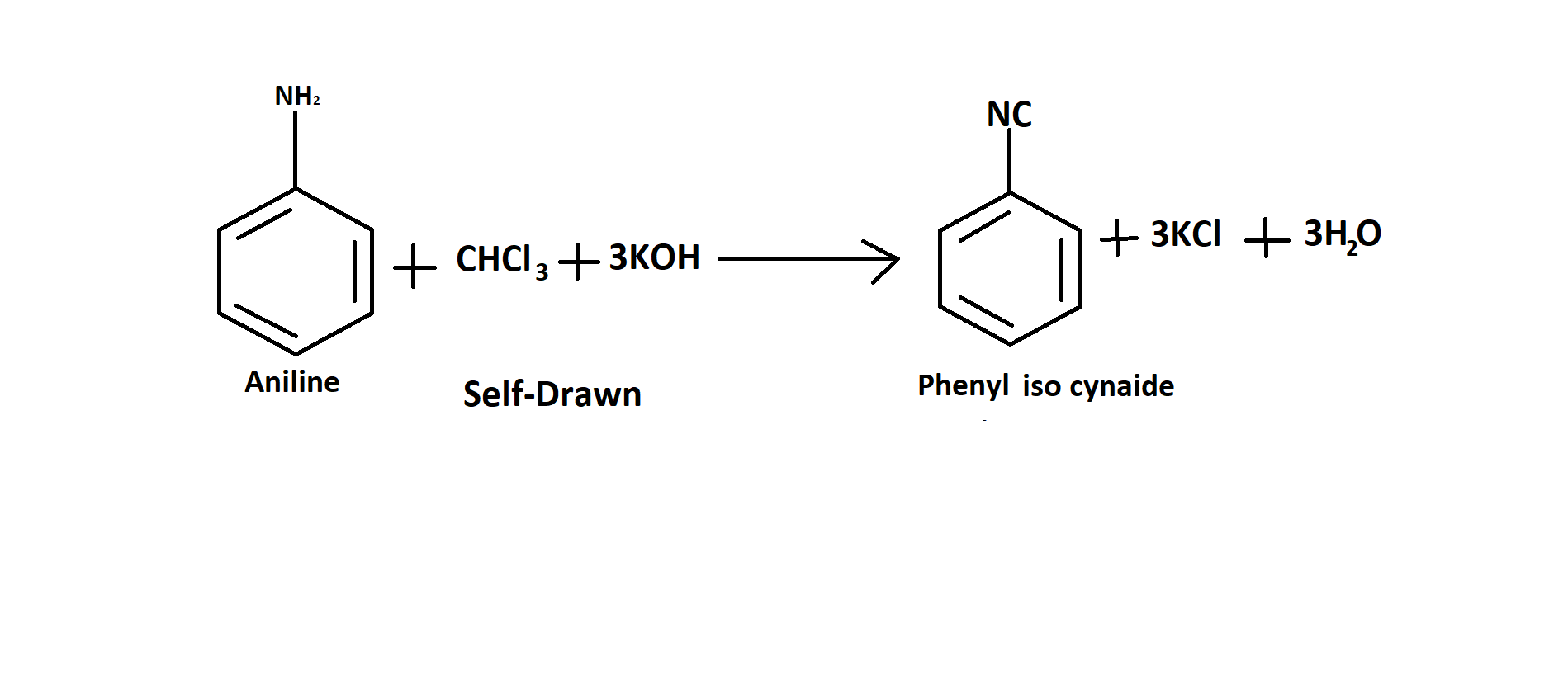
Now let's see the mechanism of the reaction. Chloroform is the first dehydrohalogenation (hydrogen halide is removed from a specific substrate) to produce dichlorocarbene intermediate. This intermediate of the dichlorocarbene is very reactive. The main amine's nucleophilic nitrogen is attacked by the electrophilic dichlorocarbene. Isonitrile is created as a result of the hydrochloric acid being eliminated. Below is a diagram that shows how the carbylamine reaction works.

So, option b is the correct option.
Note: The reaction of Chloroform with aniline and alcoholic KOH is called isocyanide test because the product is an iso-cyanide. Here, the product is an isocyanide that iso-phenyl cyanide. Only, primary aliphatic and aromatic amines provide it.
Complete step-by-step solution
The reaction of Chloroform with aniline and alcoholic KOH is called isocyanide test because the product is an iso-cyanide. Let’s know something more about the isocyanide test.
Aniline is an aromatic amine. It is a precursor to various drugs, dyes, and plastics. It is also a pollutant. Aniline is toxic, and it can be harmful to the environment.
The isocyanide test can be used to determine the presence of aniline in a sample. The test uses a reagent that reacts with aniline to form a blue colour. The reaction is not specific for aniline, so other compounds that contain aniline-like structures can also produce a blue colour. Only, primary aliphatic and aromatic amines provide it.
The isocyanide test is a quantitative test. The test can be used to determine the concentration of aniline in a sample. The test is also sensitive to low concentrations of aniline.
Now let’s see the product of the reaction of aniline and chloroform with alcoholic KOH. Obviously, the product is an isocyanide that iso-phenyl cyanide. Let's see the reaction now.

Now let's see the mechanism of the reaction. Chloroform is the first dehydrohalogenation (hydrogen halide is removed from a specific substrate) to produce dichlorocarbene intermediate. This intermediate of the dichlorocarbene is very reactive. The main amine's nucleophilic nitrogen is attacked by the electrophilic dichlorocarbene. Isonitrile is created as a result of the hydrochloric acid being eliminated. Below is a diagram that shows how the carbylamine reaction works.

So, option b is the correct option.
Note: The reaction of Chloroform with aniline and alcoholic KOH is called isocyanide test because the product is an iso-cyanide. Here, the product is an isocyanide that iso-phenyl cyanide. Only, primary aliphatic and aromatic amines provide it.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




