$CH_3−CH=CH−CH_2−CN \xrightarrow[{{H}_{2}}O]{DIBALH} Product$. Then the product is:
Answer
265.2k+ views
Hint: DIBALH (Di-isobutyl Aluminium Hydride) is a strong, bulky reducing agent which is used for the partial reduction of esters. It reduces esters to aldehydes. Nitriles are typically converted to aldehydes using the hydride reagent DIBAL-H as well.
Complete step by step solution:
The reaction of conversion of nitrile to aldehyde begins with the formation of an iminium anion by adding a hydride to the C-N triple bond. The formed imine salt then hydrolysed to form imine.
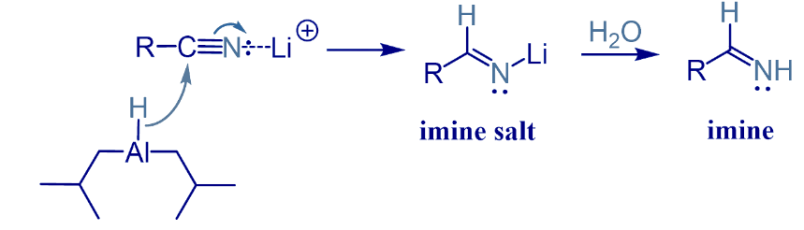
After the first hydride addition, hydrolysis of the imine to amine takes place through the nucleophilic attack of H2O.
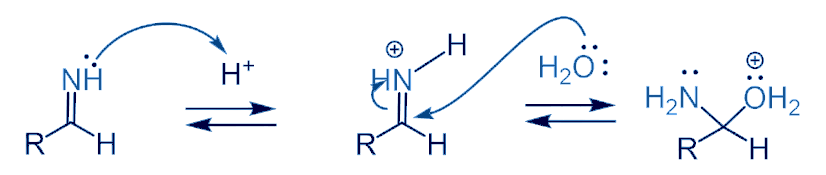
The Amine group leaves the substrate when OH attacks it as a nucleophile and forms a double bond. In the last step H2O takes out the extra H from the substrate and an aldehyde is formed.
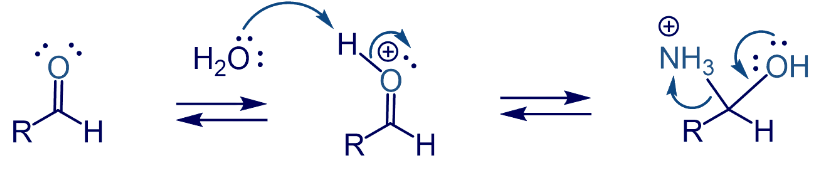
The product is CH3−CH=CH−CH2−CHO.
Additional Information: According to the suggested mechanism, DIBAL joins the nitrile through the creation of an N-Al link to form a Lewis acid-base adduct. The hydride is subsequently transferred to the nitrile's carbon. Aldehyde and ammonia are produced during aqueous workup as desired.
Note: In contrast to lithium aluminium hydride, Di-isobutyl Aluminium Hydride (DIBAL H) won't reduce the aldehyde anymore with the addition of just one equivalent. Other carbonyl substances including amides, aldehydes, ketones, and nitriles will also be reduced by it. DIBAL is most notable for what it does not do, much like Lindlar's catalyst is. It lowers esters but doesn't get all the way to alcohol; it just stops at aldehyde. The temperature is kept very low(-70° C) for the reaction to keep the reactivity in check.
Complete step by step solution:
The reaction of conversion of nitrile to aldehyde begins with the formation of an iminium anion by adding a hydride to the C-N triple bond. The formed imine salt then hydrolysed to form imine.

After the first hydride addition, hydrolysis of the imine to amine takes place through the nucleophilic attack of H2O.

The Amine group leaves the substrate when OH attacks it as a nucleophile and forms a double bond. In the last step H2O takes out the extra H from the substrate and an aldehyde is formed.

The product is CH3−CH=CH−CH2−CHO.
Additional Information: According to the suggested mechanism, DIBAL joins the nitrile through the creation of an N-Al link to form a Lewis acid-base adduct. The hydride is subsequently transferred to the nitrile's carbon. Aldehyde and ammonia are produced during aqueous workup as desired.
Note: In contrast to lithium aluminium hydride, Di-isobutyl Aluminium Hydride (DIBAL H) won't reduce the aldehyde anymore with the addition of just one equivalent. Other carbonyl substances including amides, aldehydes, ketones, and nitriles will also be reduced by it. DIBAL is most notable for what it does not do, much like Lindlar's catalyst is. It lowers esters but doesn't get all the way to alcohol; it just stops at aldehyde. The temperature is kept very low(-70° C) for the reaction to keep the reactivity in check.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




