Bottles containing \[{C_6}{H_5}I\] and \[{C_6}{H_5}C{H_2}I\] lost their original labels. They are labelled A and B for testing. A and B were separately taken in a test tube and boiled with \[NaOH\] solution. The end solution in each tube was made acidic with dilute \[HN{O_3}\] and then some \[AgN{O_3}\] solution was added. Substance B gave a yellow precipitate. Which one of the following statements is true for this experiment?
A. A was \[{C_6}{H_5}I\]
B. A was \[{C_6}{H_5}C{H_2}I\]
C. B was \[{C_6}{H_5}I\]
D. Addition of \[HN{O_3}\]was unnecessary
Answer
246k+ views
Hint: Reacting iodobenzene (\[{C_6}{H_5}I\]) and benzyl iodide (\[{C_6}{H_5}C{H_2}I\]) with sodium hydroxide (\[NaOH\]) solution results in the nucleophilic substitution reaction (via the\[{S_N}1\]pathway) of only one of them. That will be the compound giving yellow precipitate with silver nitrate (\[AgN{O_3}\]).
Complete Step by Step Solution:
The given compounds are iodobenzene (\[{C_6}{H_5}I\]) and benzyl iodide (\[{C_6}{H_5}C{H_2}I\]). Their structures are given below:
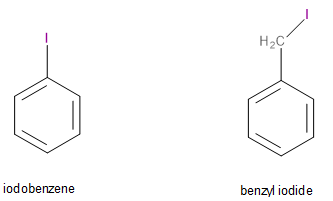
Image: Structures of iodobenzene and benzyl iodide
On boiling benzyl iodide with sodium hydroxide solution, the iodine atom gets substituted by a hydroxyl group (\[ - OH\]). This is a nucleophilic substitution reaction that takes place via the\[{S_N}1\]mechanism.
The first step involves the iodine atom leaving the molecule as an iodide ion (\[{I^ - }\]) leaving behind a benzyl carbocation as shown below:
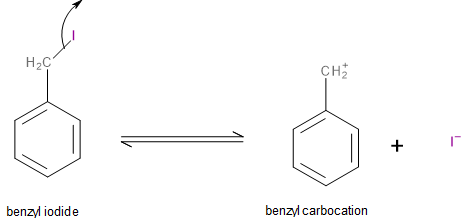
Image: Step 1
The second step involves the nucleophilic attack of the hydroxide ion (\[O{H^ - }\]) from sodium hydroxide.
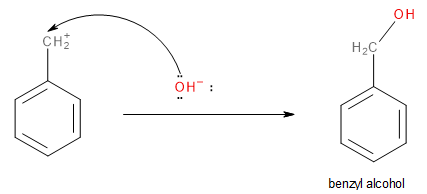
Image: Step 2
The iodide ion liberated in step 1 reacts with silver nitrate (\[AgN{O_3}\]) solution to form a pale-yellow precipitate of silver iodide (\[AgI\]).
\[{I^ - }(aq) + AgN{O_3}(aq) \to AgI(s) + NO_3^ - (aq)\]
This process does not occur in the case of iodobenzene because the carbocation formed in this case is an unstable phenyl carbocation. It is unstable because the positive charge exists on a doubly bonded, \[s{p^2}\]hybridised carbon. Carbocations become stabilised when they are attached to groups that can donate an electron pair into their empty p-orbitals. In the case of phenyl carbocation, the\[s{p^2}\]nature of the positively charged carbon atom prevents it from donating an electron pair and stabilising the carbocation.
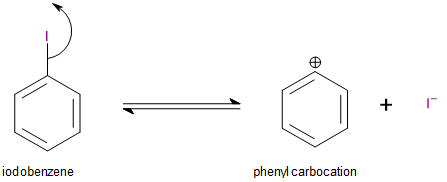
Image: Dissociation of iodobenzene to form a phenyl carbocation
On the other hand, the benzyl carbocation formed in the case of benzyl chloride is resonance stabilised. Thus, it can undergo the\[{S_N}1\]reaction with\[NaOH\].

Image: Resonating structures of benzyl carbocation
Thus, iodobenzene (\[{C_6}{H_5}I\]) is compound A and benzyl iodide (\[{C_6}{H_5}C{H_2}I\]) is compound B.
Thus, option A is correct.
Note: The answer to this question entirely hinges on the various factors that promote or inhibit\[{S_N}1\]reactions such as the stability of carbocations. Addition of dilute \[HN{O_3}\]also factors into this since\[HN{O_3}\]is protic and we know that protic environments promote\[{S_N}1\]reactions by helping make the carbocation intermediate stable.
Complete Step by Step Solution:
The given compounds are iodobenzene (\[{C_6}{H_5}I\]) and benzyl iodide (\[{C_6}{H_5}C{H_2}I\]). Their structures are given below:

Image: Structures of iodobenzene and benzyl iodide
On boiling benzyl iodide with sodium hydroxide solution, the iodine atom gets substituted by a hydroxyl group (\[ - OH\]). This is a nucleophilic substitution reaction that takes place via the\[{S_N}1\]mechanism.
The first step involves the iodine atom leaving the molecule as an iodide ion (\[{I^ - }\]) leaving behind a benzyl carbocation as shown below:

Image: Step 1
The second step involves the nucleophilic attack of the hydroxide ion (\[O{H^ - }\]) from sodium hydroxide.

Image: Step 2
The iodide ion liberated in step 1 reacts with silver nitrate (\[AgN{O_3}\]) solution to form a pale-yellow precipitate of silver iodide (\[AgI\]).
\[{I^ - }(aq) + AgN{O_3}(aq) \to AgI(s) + NO_3^ - (aq)\]
This process does not occur in the case of iodobenzene because the carbocation formed in this case is an unstable phenyl carbocation. It is unstable because the positive charge exists on a doubly bonded, \[s{p^2}\]hybridised carbon. Carbocations become stabilised when they are attached to groups that can donate an electron pair into their empty p-orbitals. In the case of phenyl carbocation, the\[s{p^2}\]nature of the positively charged carbon atom prevents it from donating an electron pair and stabilising the carbocation.

Image: Dissociation of iodobenzene to form a phenyl carbocation
On the other hand, the benzyl carbocation formed in the case of benzyl chloride is resonance stabilised. Thus, it can undergo the\[{S_N}1\]reaction with\[NaOH\].

Image: Resonating structures of benzyl carbocation
Thus, iodobenzene (\[{C_6}{H_5}I\]) is compound A and benzyl iodide (\[{C_6}{H_5}C{H_2}I\]) is compound B.
Thus, option A is correct.
Note: The answer to this question entirely hinges on the various factors that promote or inhibit\[{S_N}1\]reactions such as the stability of carbocations. Addition of dilute \[HN{O_3}\]also factors into this since\[HN{O_3}\]is protic and we know that protic environments promote\[{S_N}1\]reactions by helping make the carbocation intermediate stable.
Recently Updated Pages
Which of the following contains the highest percentage class 12 chemistry JEE_Main

Protein can be most easily removed from A Alkanes B class 12 chemistry JEE_Main

PhCOCHBr2xrightarrowOHAxrightarrowOHBxrightarrowH+C class 12 chemistry JEE_Main

When alcohol reacts with conc H2SO4 intermediate compound class 12 chemistry JEE_Main

A nanopeptide contains peptide linkages A 10 B 8 C class 12 chemistry JEE_Main

Which one of the following is paramagnetic A NO B N2O class 12 chemistry JEE_Main

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Collisions: Types and Examples for Students

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The D And F Block Elements - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




