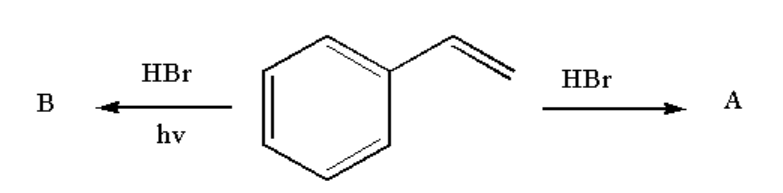
Analyse the following reaction and identify the nature of A and B.
Answer
255.9k+ views
Hint: The chemical name of HBr is hydrogen bromide. In the given reaction it should be noted that one reaction is taking place in presence of light and other in the absence of light. The reaction taking place in the presence of light forms free radicals.
Complete Step by Step Solution:
The reaction of a given compound with hydrogen bromide is an electrophilic addition reaction.
In an electrophilic addition reaction, the substrate is first attacked by an electrophile which results in the addition of an atom or molecule across multiple bonds.
In the first step, hydrogen bromide breaks to form hydrogen ion and bromide ion.
The reaction is shown below.
\[HBr \to {H^ + } + B{r^ - }\]
Image: Step 1: Formation of electrophile
In the second step, the hydrogen ion attack the double bond and a carbocation complex is formed.
The reaction is shown below.
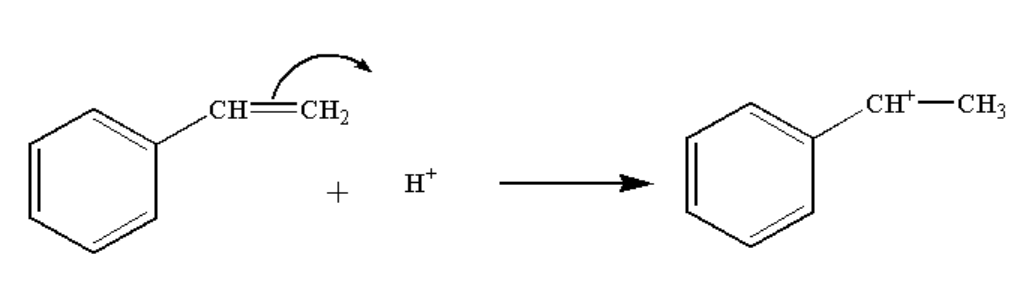
Image: Step 2: Formation of carbocation
In the third step, the bromide ion attacks the carbocation to form the compound A.
The reaction is shown below.
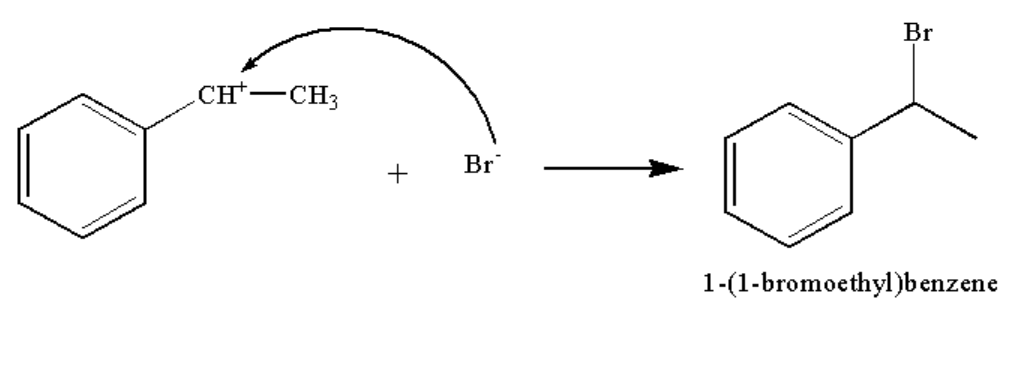
Image: Step 3: Formation of compound A
The reaction of a given compound with hydrogen bromide in presence of light is a free radical addition reaction.
Free radical addition reaction involves the addition of free radicals. Free radicals are formed by homolytic cleavage.
The hydrogen bromide undergoes homolysis to form a free radical of hydrogen and bromine.
The reaction is shown below.
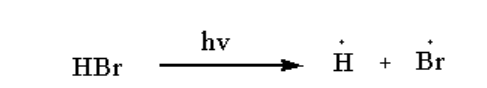
Image: Step 1: Free radical formation
Next, bromine radical is added to the compound
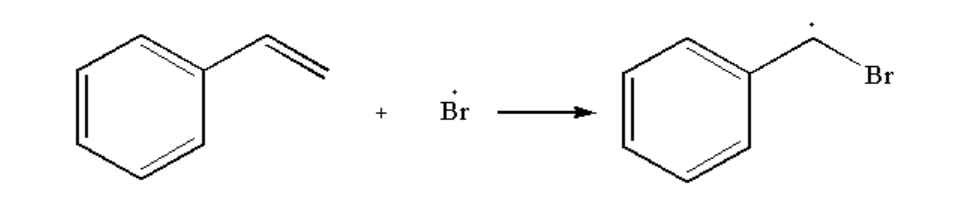
Image: Step 2
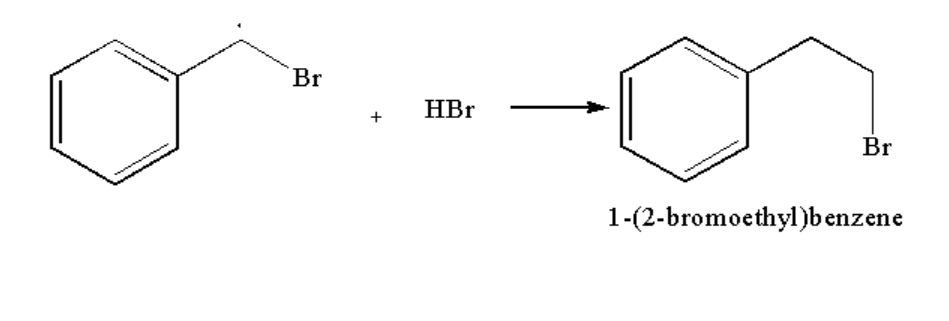
Image: Step 3: Formation of compound B
Therefore compound A is 1-(1-bromoethylbenzene) and compound B is 1-(2-bromoethyl)benzene.
Note: The anti-Markovnikov free radical addition of haloalkane only takes place in HBr in presence of hydrogen peroxide. Hydrogen peroxide starts the chain reaction in the initiation step..
Complete Step by Step Solution:
The reaction of a given compound with hydrogen bromide is an electrophilic addition reaction.
In an electrophilic addition reaction, the substrate is first attacked by an electrophile which results in the addition of an atom or molecule across multiple bonds.
In the first step, hydrogen bromide breaks to form hydrogen ion and bromide ion.
The reaction is shown below.
\[HBr \to {H^ + } + B{r^ - }\]
Image: Step 1: Formation of electrophile
In the second step, the hydrogen ion attack the double bond and a carbocation complex is formed.
The reaction is shown below.

Image: Step 2: Formation of carbocation
In the third step, the bromide ion attacks the carbocation to form the compound A.
The reaction is shown below.

Image: Step 3: Formation of compound A
The reaction of a given compound with hydrogen bromide in presence of light is a free radical addition reaction.
Free radical addition reaction involves the addition of free radicals. Free radicals are formed by homolytic cleavage.
The hydrogen bromide undergoes homolysis to form a free radical of hydrogen and bromine.
The reaction is shown below.

Image: Step 1: Free radical formation
Next, bromine radical is added to the compound

Image: Step 2

Image: Step 3: Formation of compound B
Therefore compound A is 1-(1-bromoethylbenzene) and compound B is 1-(2-bromoethyl)benzene.
Note: The anti-Markovnikov free radical addition of haloalkane only takes place in HBr in presence of hydrogen peroxide. Hydrogen peroxide starts the chain reaction in the initiation step..
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




