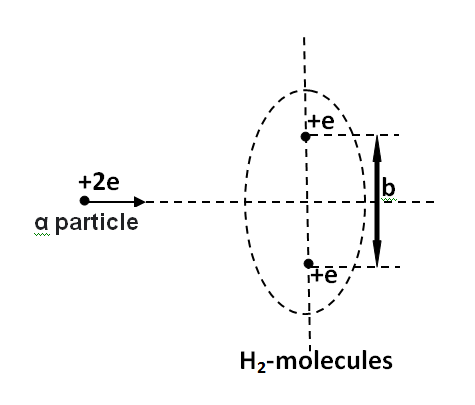
An \[\alpha \] particle passes rapidly through the exact centre of a hydrogen molecule, moving on a line perpendicular to the internuclear axis. The distance between the nuclei is \[b\] . Where on its path does the \[\alpha \] -particle experience the greatest force? Assume that the nuclei do not move much during the passage of the \[\alpha \] particle. Also neglect the electric field of the electrons in the molecule.
A. $\dfrac{b}{2}$
B. $\dfrac{b}{{2\sqrt 2 }}$
C. $\dfrac{b}{{\sqrt 2 }}$
D. None of these
Answer
266.7k+ views
Hint: To solve this question, we will first consider a point on the perpendicular bisector of the line joining the two hydrogen atoms. To find the force between the \[\alpha \] particle and the hydrogen atom, we will consider the \[\alpha \] particle at that point and write the expression of force. Now, we know that the condition to find the extreme value of a function whose first derivative is kept equal to zero. We will use this concept to get the value of the greatest force experienced by the \[\alpha \] particle.
Formula Used:
Force between two charges,
$F = k\dfrac{{{q_1}{q_2}}}{{{r^2}}}$
where ${q_1}\,\& \,{q_2}$ are the two charges and $r$ is the distance between them.
Also, $k = \dfrac{1}{{4\pi { \in _o}}}$
Quotient formula of differentiation,
$\dfrac{d}{{dx}}\left( {\dfrac{{f\left( x \right)}}{{g\left( x \right)}}} \right) = \dfrac{{f'\left( x \right)g\left( x \right) - f\left( x \right)g'\left( x \right)}}{{{{\left( {g\left( x \right)} \right)}^2}}}$
Complete step by step solution:
Given: Charge on a hydrogen atom, ${q_1} = e$
Charge on the \[\alpha \] particles, ${q_2} = 2e$
Distance between the nuclei \[ = b\]
Let $F$ be the force between the \[\alpha \] particle and the hydrogen atom. Draw a perpendicular bisector of the line joining the nuclei of the two hydrogen atoms. Consider a point $P$ on it at a distance \[x\] from the foot of the perpendicular.
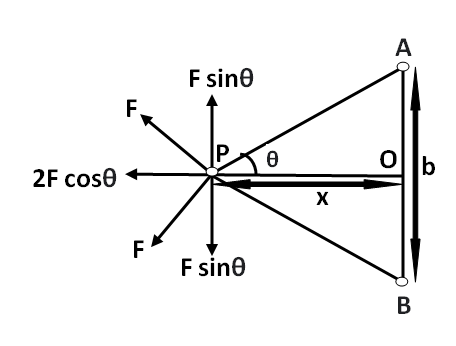
From the diagram above we can see that the vertical components of the force $F$ cancel each other. Hence, the net force becomes,
${F_{net}} = 2F\cos \theta $
Where $\cos \theta = \dfrac{x}{{{{\left( {{x^2} + {{\left( {\dfrac{b}{2}} \right)}^2}} \right)}^{\dfrac{1}{2}}}}}$
(denominator is obtained using Pythagoras theorem in $\vartriangle AOP$ )
This implies,
\[{F_{net}} = 2F\dfrac{x}{{{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}}}}\] ...(1)
Also,
$F = k\dfrac{{{q_1}{q_2}}}{{{r^2}}}$
That is,
$F = k\dfrac{{\left( e \right)\left( {2e} \right)}}{{{{\left( {{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}}} \right)}^2}}} \\ $...(2)
Substituting equation (2) in equation (1), we get,
\[{F_{net}} = 2\left( {k\dfrac{{\left( e \right)\left( {2e} \right)}}{{{{\left( {{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}}} \right)}^2}}}} \right)\dfrac{x}{{{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}}}} \\ \]
Simplifying this, we get,
\[{F_{net}} = \dfrac{{4kx{e^2}}}{{{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{3}{2}}}}}\]
Now, we know that the condition of extremum is: $\dfrac{{d{F_{net}}}}{{dx}} = 0$ , that is, the first derivative is kept equal to zero to find the extreme value of a function.
Using quotient formula we get,
$\dfrac{{d{F_{net}}}}{{dx}} = 4k{e^2}\dfrac{{\left( {1 \times {{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{3}{2}}} - \left( {x \times \dfrac{3}{2}{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}} \times 2x} \right)} \right)}}{{{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^3}}} \\ $
Putting this equal to zero, we get,
$4k{e^2}\dfrac{{\left( {1 \times {{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{3}{2}}} - \left( {x \times \dfrac{3}{2}{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}} \times 2x} \right)} \right)}}{{{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^3}}} = 0$
Simplifying this, we get,
$\left( {1 \times {{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{3}{2}}} - \left( {x \times \dfrac{3}{2}{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}} \times 2x} \right)} \right) = 0 \\ $
This implies,
$\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right){\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)^{\dfrac{1}{2}}} = 3{x^2}{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)^{\dfrac{1}{2}}} \\ $
Solving this, we get,
$\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right) = 3{x^2} \\ $
This gives, $2{x^2} = \dfrac{{{b^2}}}{4} \\ $
Hence, $x = \pm \dfrac{b}{{2\sqrt 2 }}$
Thus, the $\alpha $ particle experience greatest force at a distance $x = \pm \dfrac{b}{{2\sqrt 2 }}$ from the foot of the perpendicular bisector of the line joining the nuclei of the two hydrogen atoms.
Hence, option B is the correct answer.
Note: Before doing this question one must know the rules of differentiation. Also, an important point to note in this question is that the direct expression of force between the $\alpha $ particle and a hydrogen atom is not the net force. We need to take into consideration the components of this force.
Formula Used:
Force between two charges,
$F = k\dfrac{{{q_1}{q_2}}}{{{r^2}}}$
where ${q_1}\,\& \,{q_2}$ are the two charges and $r$ is the distance between them.
Also, $k = \dfrac{1}{{4\pi { \in _o}}}$
Quotient formula of differentiation,
$\dfrac{d}{{dx}}\left( {\dfrac{{f\left( x \right)}}{{g\left( x \right)}}} \right) = \dfrac{{f'\left( x \right)g\left( x \right) - f\left( x \right)g'\left( x \right)}}{{{{\left( {g\left( x \right)} \right)}^2}}}$
Complete step by step solution:
Given: Charge on a hydrogen atom, ${q_1} = e$
Charge on the \[\alpha \] particles, ${q_2} = 2e$
Distance between the nuclei \[ = b\]
Let $F$ be the force between the \[\alpha \] particle and the hydrogen atom. Draw a perpendicular bisector of the line joining the nuclei of the two hydrogen atoms. Consider a point $P$ on it at a distance \[x\] from the foot of the perpendicular.

From the diagram above we can see that the vertical components of the force $F$ cancel each other. Hence, the net force becomes,
${F_{net}} = 2F\cos \theta $
Where $\cos \theta = \dfrac{x}{{{{\left( {{x^2} + {{\left( {\dfrac{b}{2}} \right)}^2}} \right)}^{\dfrac{1}{2}}}}}$
(denominator is obtained using Pythagoras theorem in $\vartriangle AOP$ )
This implies,
\[{F_{net}} = 2F\dfrac{x}{{{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}}}}\] ...(1)
Also,
$F = k\dfrac{{{q_1}{q_2}}}{{{r^2}}}$
That is,
$F = k\dfrac{{\left( e \right)\left( {2e} \right)}}{{{{\left( {{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}}} \right)}^2}}} \\ $...(2)
Substituting equation (2) in equation (1), we get,
\[{F_{net}} = 2\left( {k\dfrac{{\left( e \right)\left( {2e} \right)}}{{{{\left( {{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}}} \right)}^2}}}} \right)\dfrac{x}{{{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}}}} \\ \]
Simplifying this, we get,
\[{F_{net}} = \dfrac{{4kx{e^2}}}{{{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{3}{2}}}}}\]
Now, we know that the condition of extremum is: $\dfrac{{d{F_{net}}}}{{dx}} = 0$ , that is, the first derivative is kept equal to zero to find the extreme value of a function.
Using quotient formula we get,
$\dfrac{{d{F_{net}}}}{{dx}} = 4k{e^2}\dfrac{{\left( {1 \times {{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{3}{2}}} - \left( {x \times \dfrac{3}{2}{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}} \times 2x} \right)} \right)}}{{{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^3}}} \\ $
Putting this equal to zero, we get,
$4k{e^2}\dfrac{{\left( {1 \times {{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{3}{2}}} - \left( {x \times \dfrac{3}{2}{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}} \times 2x} \right)} \right)}}{{{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^3}}} = 0$
Simplifying this, we get,
$\left( {1 \times {{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{3}{2}}} - \left( {x \times \dfrac{3}{2}{{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)}^{\dfrac{1}{2}}} \times 2x} \right)} \right) = 0 \\ $
This implies,
$\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right){\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)^{\dfrac{1}{2}}} = 3{x^2}{\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right)^{\dfrac{1}{2}}} \\ $
Solving this, we get,
$\left( {{x^2} + \dfrac{{{b^2}}}{4}} \right) = 3{x^2} \\ $
This gives, $2{x^2} = \dfrac{{{b^2}}}{4} \\ $
Hence, $x = \pm \dfrac{b}{{2\sqrt 2 }}$
Thus, the $\alpha $ particle experience greatest force at a distance $x = \pm \dfrac{b}{{2\sqrt 2 }}$ from the foot of the perpendicular bisector of the line joining the nuclei of the two hydrogen atoms.
Hence, option B is the correct answer.
Note: Before doing this question one must know the rules of differentiation. Also, an important point to note in this question is that the direct expression of force between the $\alpha $ particle and a hydrogen atom is not the net force. We need to take into consideration the components of this force.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Young’s Double Slit Experiment Derivation Explained

Wheatstone Bridge – Principle, Formula, Diagram & Applications

Circuit Switching vs Packet Switching: Key Differences Explained

Mass vs Weight: Key Differences Explained for Students

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Kinematics Mock Test for JEE Main 2025-26: Practice & Ace the Exam

Kinematics Mock Test for JEE Main 2025-26: Comprehensive Practice

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Other Pages
CBSE Class 12 Physics Question Paper 2026: Download SET-wise PDF with Answer Key & Analysis

JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Derivation of Equation of Trajectory Explained for Students




