Ammonium acetate reacts with acetic acid at \[110^\circ {\rm{C}}\] to form
A. Acetamide
B. Formamide
C. Ammonium cyanate
D. Urea
Answer
248.7k+ views
Hint: Ammonium acetate is an organic chemical compound. Its appearance is a crystalline solid of white colour. Its odour is acetous-like. This is the salt prepared by the reaction of acetic acid and ammonia.
Complete step by step solution:
Let’s draw the structure of ammonium acetate first.
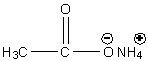
Image: Ammonium acetate
Now, we have to understand the reaction of ammonium acetate with acetic acid at 110 degrees Celsius. When ammonium acetate undergoes a reaction with acetic acid, a loss of water molecules takes place. The chemical reaction is,
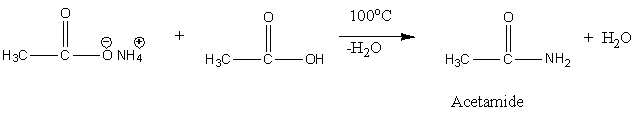
Image: Reaction of ammonium acetate and acetic acid.
Therefore, the reaction of ammonium acetate and acetic acid gives acetamide.
Hence, the option A is right.
Additional Information:
The general name of the compound ethanamide is acetamide. It is chemically represented as\[{\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{CON}}{{\rm{H}}_{\rm{2}}}\] . It is prepared from ethanoic acid. In laboratory preparation, it is prepared from ammonium acetate via the process of dehydration. It smells like ammonia or vinegar.
Note: There is easy solubility of acetamide in water, benzene and chloroform. It belongs to the class of amides that is obtained from the condensation of acetic acid with ammonia. In nature, it is found in beetroot. Some of the uses of acetamides are explosive making, stabiliser, hygroscopic agent etc. It is a substance of combustible nature. The heating of acetamide produces a toxic gas. Exposure to Acetamide is dangerous to health.
Complete step by step solution:
Let’s draw the structure of ammonium acetate first.

Image: Ammonium acetate
Now, we have to understand the reaction of ammonium acetate with acetic acid at 110 degrees Celsius. When ammonium acetate undergoes a reaction with acetic acid, a loss of water molecules takes place. The chemical reaction is,
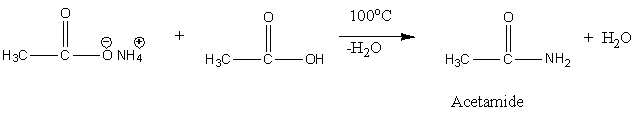
Image: Reaction of ammonium acetate and acetic acid.
Therefore, the reaction of ammonium acetate and acetic acid gives acetamide.
Hence, the option A is right.
Additional Information:
The general name of the compound ethanamide is acetamide. It is chemically represented as\[{\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{CON}}{{\rm{H}}_{\rm{2}}}\] . It is prepared from ethanoic acid. In laboratory preparation, it is prepared from ammonium acetate via the process of dehydration. It smells like ammonia or vinegar.
Note: There is easy solubility of acetamide in water, benzene and chloroform. It belongs to the class of amides that is obtained from the condensation of acetic acid with ammonia. In nature, it is found in beetroot. Some of the uses of acetamides are explosive making, stabiliser, hygroscopic agent etc. It is a substance of combustible nature. The heating of acetamide produces a toxic gas. Exposure to Acetamide is dangerous to health.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Derivation of Equation of Trajectory Explained for Students

Understanding the Angle of Deviation in a Prism

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The D And F Block Elements - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




