A reaction of ethyl amine and acetic anhydride leads to the formation of:
(A) $C{{H}_{3}}NHCOC{{H}_{3}}$
(B) ${{C}_{2}}{{H}_{5}}CONHC{{H}_{3}}$
(C) $C{{H}_{3}}CONH{{C}_{2}}{{H}_{5}}$
(D) $C{{H}_{3}}-CH=NO{{C}_{2}}{{H}_{5}}$
Answer
264.6k+ views
Hint: A reaction of ethyl amine and acetic anhydride is an acetylation reaction. An acetylation reaction is the reaction in which an acetyl group is introduced into a compound. This reaction can occur by the addition of acetic anhydride or acetyl chloride. It is a nucleophilic substitution reaction. This reaction is also known as ethanoylation.
Complete Step by Step Answer:
The ethyl amine consists of an ethyl group attached to a $N{{H}_{2}}$ group. When the acetylation reaction of ethyl amine takes place by the addition of acetic anhydride, N-ethyl acetamide is formed. The chemical formula of N-ethyl acetamide is $C{{H}_{3}}CONH{{C}_{2}}{{H}_{5}}$. Nitrogen atoms of ethyl amine are acetylated in this reaction. In this reaction, one of the hydrogen atoms of the $N{{H}_{2}}$ group is replaced by an acetyl group.
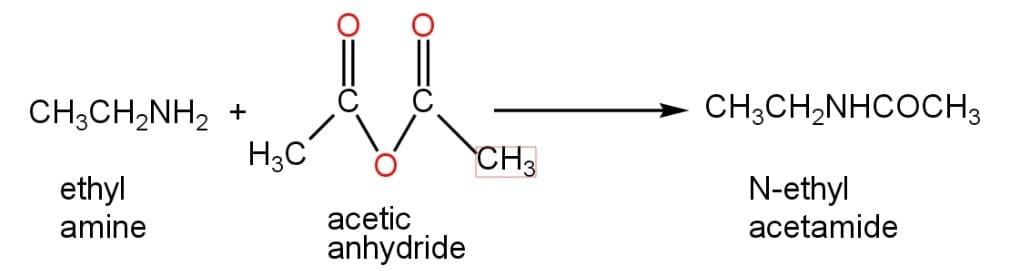
Correct Option: (C)$C{{H}_{3}}CONH{{C}_{2}}{{H}_{5}}$.
Additional Information: Many compounds like aspirin and proteins can be synthesised with the help of an acetylation reaction. These reactions play a crucial role in drug biotransformation, which is the acetylation reaction-aided processing and elimination of medicines by the body. The inverse reaction, which is the reaction in which the acetyl group is removed completely, is known as the deacetylation reaction.
Note: Acetylation is a modification that can significantly modify a protein's function by changing its hydrophobicity, solubility, and surface characteristics. These changes may have an impact on the protein's conformation and interactions with substrates, cofactors, and other macromolecules.
Complete Step by Step Answer:
The ethyl amine consists of an ethyl group attached to a $N{{H}_{2}}$ group. When the acetylation reaction of ethyl amine takes place by the addition of acetic anhydride, N-ethyl acetamide is formed. The chemical formula of N-ethyl acetamide is $C{{H}_{3}}CONH{{C}_{2}}{{H}_{5}}$. Nitrogen atoms of ethyl amine are acetylated in this reaction. In this reaction, one of the hydrogen atoms of the $N{{H}_{2}}$ group is replaced by an acetyl group.

Correct Option: (C)$C{{H}_{3}}CONH{{C}_{2}}{{H}_{5}}$.
Additional Information: Many compounds like aspirin and proteins can be synthesised with the help of an acetylation reaction. These reactions play a crucial role in drug biotransformation, which is the acetylation reaction-aided processing and elimination of medicines by the body. The inverse reaction, which is the reaction in which the acetyl group is removed completely, is known as the deacetylation reaction.
Note: Acetylation is a modification that can significantly modify a protein's function by changing its hydrophobicity, solubility, and surface characteristics. These changes may have an impact on the protein's conformation and interactions with substrates, cofactors, and other macromolecules.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




