



List of Important Salts and Their Uses with Examples and Formulas
Salts are ionic compounds formed when an acid reacts with a base in a neutralization reaction. They play a vital role in chemistry, daily life, medicine, agriculture, and industries. From common table salt used in food to chemicals used in fertilizers, construction, and medical treatments, salts are essential substances that support human life and industrial development. Understanding the important salts and their uses is crucial for students, especially for competitive exams like NEET, where questions are frequently asked from basic chemistry and everyday applications.
What Are Salts?
A salt is a chemical compound formed by the complete or partial replacement of hydrogen ions of an acid by a metal or ammonium ion. Salts are generally formed through a neutralization reaction between an acid and a base. For example, when hydrochloric acid reacts with sodium hydroxide, sodium chloride and water are produced.
- Salts are usually crystalline solids.
- They have high melting and boiling points.
- Many salts are soluble in water.
- They conduct electricity in molten or aqueous state.
Classification of Salts
1. Neutral Salts
Formed by complete neutralization of a strong acid and a strong base. Example - Sodium chloride.
2. Acidic Salts
Formed when a strong acid reacts with a weak base. Example - Ammonium chloride.
3. Basic Salts
Formed when a strong base reacts with a weak acid. Example - Sodium carbonate.
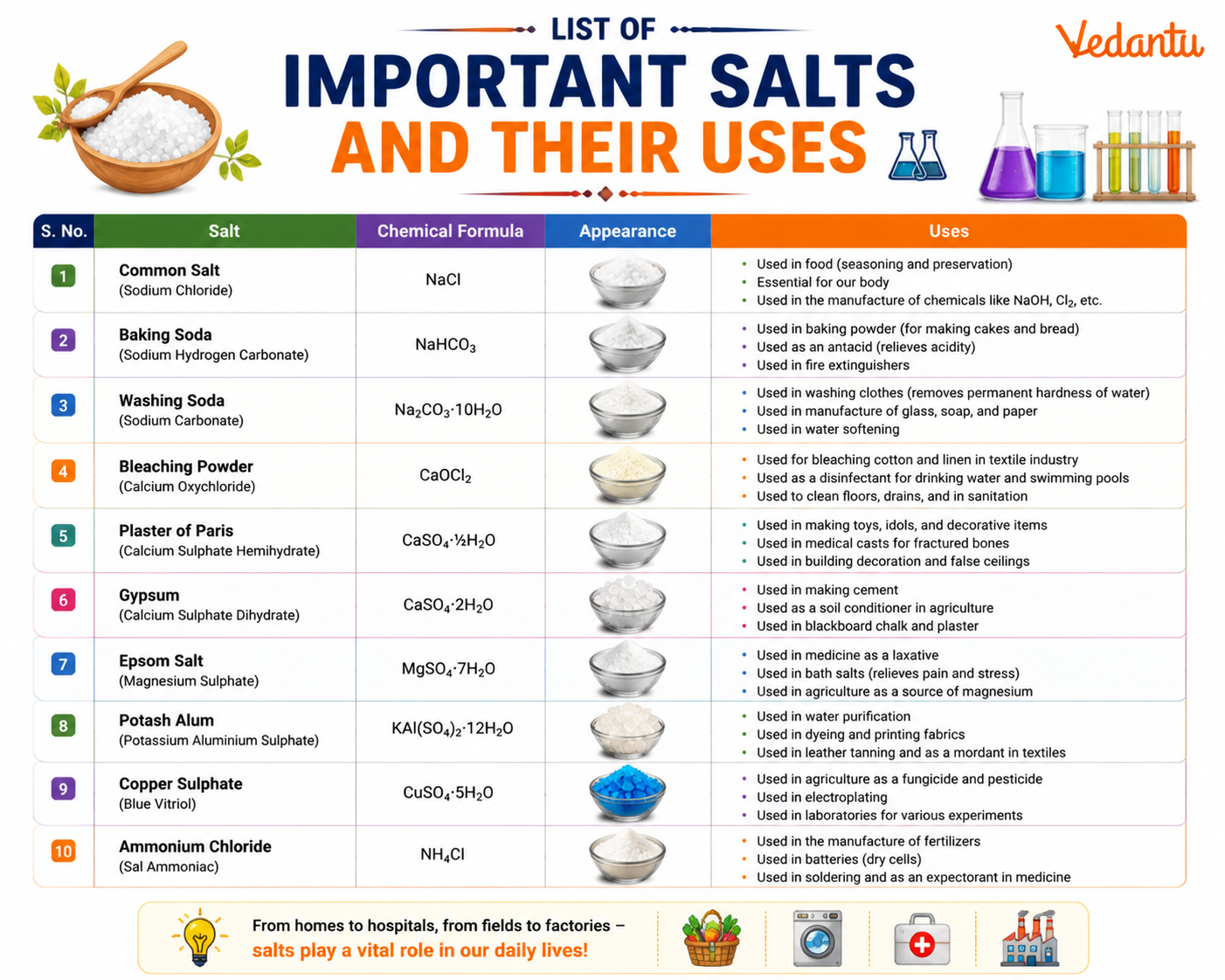
List of Important Salts and Their Uses
| Name of Salt | Chemical Formula | Major Uses |
|---|---|---|
| Common Salt | NaCl | Cooking, food preservation, raw material for chemicals |
| Baking Soda | NaHCO3 | Baking, antacid, fire extinguisher |
| Washing Soda | Na2CO3 | Cleaning agent, water softening, glass making |
| Plaster of Paris | CaSO4·1/2H2O | Moulds, casts, decorative items |
| Bleaching Powder | CaOCl2 | Disinfectant, bleaching textiles, water purification |
| Blue Vitriol | CuSO4·5H2O | Fungicide, laboratory reagent, electroplating |
| Potash Alum | KAl(SO4)2·12H2O | Water purification, shaving alum, dyeing |
The above salts are frequently discussed in school chemistry and are important for competitive exams. Their preparation methods and applications are commonly tested in NEET and other entrance examinations.
Detailed Uses of Important Salts
Common Salt - Sodium Chloride
- Used in cooking and food preservation.
- Raw material for manufacturing sodium hydroxide, chlorine, and hydrochloric acid.
- Used in saline solutions in hospitals.
Baking Soda - Sodium Hydrogen Carbonate
- Used in baking as a leavening agent.
- Acts as an antacid to relieve acidity.
- Used in soda acid fire extinguishers.
Washing Soda - Sodium Carbonate
- Softens hard water by removing calcium and magnesium ions.
- Used in manufacturing glass, soap, and paper.
- Cleaning agent for domestic purposes.
Plaster of Paris
- Used in making statues and decorative materials.
- Used for making moulds and casts.
- Orthopedic casts for fractured bones.
Bleaching Powder
- Disinfects drinking water.
- Bleaches cotton and linen in textile industry.
- Used as an oxidizing agent in chemical industries.
Importance of Salts in Daily Life and Exams
Salts are indispensable in daily life. They are used in food, medicine, agriculture, water purification, and construction. In competitive exams like NEET, students must understand their chemical formulas, preparation reactions, properties, and practical applications. Questions are often conceptual and application based, requiring a clear understanding of how and why each salt is used.
Key Points for Quick Revision
- Salts are formed by neutralization reactions.
- Common salt is the most widely used salt in daily life.
- Baking soda releases carbon dioxide on heating.
- Washing soda is used for softening hard water.
- Plaster of Paris sets quickly when mixed with water.
- Bleaching powder is used for disinfection and bleaching.
A clear understanding of important salts and their uses strengthens the foundation of chemistry and helps students perform well in school exams and competitive tests. Regular revision of formulas and applications is essential for mastering this topic.
FAQs on List of Important Salts and Their Uses in Chemistry and Daily Life
1. What are salts and how are they formed?
Salts are ionic compounds formed when an acid reacts with a base in a process called neutralization reaction.
• Produced by reaction between acid + base → salt + water
• Made of positive (cation) and negative (anion) ions
• Can be acidic, basic, or neutral salts depending on their parent acid and base
• Common examples include NaCl (sodium chloride) and CuSO₄ (copper sulphate)
This concept is important in chemistry basics, chemical compounds, and competitive exams.
2. What are the important salts and their chemical formulas?
Some important salts and their formulas are commonly studied in General Knowledge (GK) and school chemistry.
• Sodium Chloride (NaCl) – Common salt
• Sodium Carbonate (Na₂CO₃) – Washing soda
• Sodium Bicarbonate (NaHCO₃) – Baking soda
• Calcium Carbonate (CaCO₃) – Limestone
• Copper Sulphate (CuSO₄) – Blue vitriol
• Potash Alum (KAl(SO₄)₂·12H₂O) – Alum
These salts are frequently asked in GK questions, science quizzes, and board exams.
3. What are the uses of sodium chloride (NaCl)?
Sodium chloride is the most commonly used salt in daily life and industry.
• Used as table salt for cooking and food preservation
• Essential for maintaining body fluid balance
• Raw material in making caustic soda, chlorine, and baking soda
• Used in saline solutions in hospitals
This salt plays a vital role in health, food industry, and chemical manufacturing.
4. What are the uses of washing soda (sodium carbonate)?
Washing soda is an important industrial salt used for cleaning and manufacturing.
• Used as a cleaning agent and water softener
• Manufacture of glass, soap, and paper
• Removes permanent hardness of water
• Used in preparation of borax and other sodium compounds
It is commonly asked in topics related to hard water treatment and chemical industries.
5. What are the uses of baking soda (sodium bicarbonate)?
Baking soda is a versatile salt widely used in cooking and medicine.
• Used in baking powder to make cakes and bread soft
• Acts as an antacid to relieve acidity
• Used in fire extinguishers (releases CO₂ gas)
• Cleaning agent for household purposes
This salt is important in food chemistry, medical uses, and safety equipment.
6. What are the uses of copper sulphate (CuSO₄)?
Copper sulphate is a blue-colored salt used mainly in agriculture and laboratories.
• Used as a fungicide and pesticide
• Preparation of Bordeaux mixture for crops
• Used in electroplating and chemical experiments
• Testing for presence of water in anhydrous form
It is significant in agriculture GK, laboratory chemicals, and industrial salts.
7. What are the uses of calcium carbonate (CaCO₃)?
Calcium carbonate is a naturally occurring salt found in rocks and shells.
• Used in manufacture of cement and lime
• Ingredient in toothpaste and chalk
• Acts as a dietary calcium supplement
• Used in construction as marble and limestone
This salt is important in construction materials, health supplements, and geology.
8. What is potash alum and what are its uses?
Potash alum is a double salt commonly used for purification and medicinal purposes.
• Used in water purification process
• Applied as an antiseptic after shaving
• Used in dyeing and tanning industries
• Stops minor bleeding due to its astringent properties
It is frequently asked in important chemical compounds and daily life chemistry.
9. What is the difference between acidic, basic, and neutral salts?
Salts are classified based on the strength of the parent acid and base.
• Neutral salts: Formed from strong acid + strong base (e.g., NaCl)
• Acidic salts: Formed from strong acid + weak base (e.g., NH₄Cl)
• Basic salts: Formed from weak acid + strong base (e.g., Na₂CO₃)
This classification is important in understanding pH value, salt hydrolysis, and chemical properties of salts.
10. Why are important salts and their uses frequently asked in GK exams?
Important salts and their uses are commonly asked because they connect chemistry with daily life and industry.
• Frequently appear in school exams and competitive exams
• Related to health, agriculture, construction, and manufacturing
• Help understand chemical reactions and practical applications
• Form part of basic science general knowledge (GK) syllabus
Knowing these salts improves performance in science quizzes, SSC, UPSC, and other entrance exams.





















