Write the reactions involved in Gabriel Phthalimide synthesis.
Answer
601.2k+ views
Hint: Let us first understand about Gabriel Phthalimide reaction. It is a chemical reaction in which the primary alkalyhallides are transformed into primary amines. Generally this reaction uses potassium phthalimide. This chemical reaction is named after the German chemist Siegmund Gabriel.
Complete Step by step answer: Gabriel Phthalimide synthesis is the chemical reaction that is involved in the preparation of amines. When potassium hydroxide reacts with phthalimide, a neulceophile in the form of an imide ion is formed. The imide ion along with alkyl halide undergoes a nucleophilic substitution reaction that forms an intermediate N-alkyl phthalimide. This intermediate phthalimide further undergoes hydrolysis or hydrazinolysis to yield a primary alkyl amine. Basically, the Gabriel reaction involves the alkylation of sulfonamides and amides that is followed by deprotection to obtain primary amines.
Steps involved in Gabriel Phthalimide synthesis:
-The potassium or sodium salts of phthalimide are N-alkylated with the primary alkyl halide to obtain the corresponding N-alkyl phthalimide.
-The phthalimide obtained by alkylation is not a nucleophile and hence does not react further. Therefore hydrolyzation of this intermediate phthalimide is necessary.
-Further, the alkyl phthalimide obtained undergoes hydrolysis to obtain primary amine.
Gabriel Phthalimide reaction:
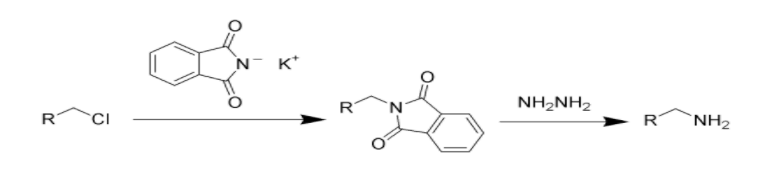
Note: The aryl amines cannot be prepared by Gabriel phthalimide synthesis because aryl halides do not undergo simple nucleophilic substitution. The huge benefit of Gabriel phthalimide synthesis is that it avoids over alkylation.
Complete Step by step answer: Gabriel Phthalimide synthesis is the chemical reaction that is involved in the preparation of amines. When potassium hydroxide reacts with phthalimide, a neulceophile in the form of an imide ion is formed. The imide ion along with alkyl halide undergoes a nucleophilic substitution reaction that forms an intermediate N-alkyl phthalimide. This intermediate phthalimide further undergoes hydrolysis or hydrazinolysis to yield a primary alkyl amine. Basically, the Gabriel reaction involves the alkylation of sulfonamides and amides that is followed by deprotection to obtain primary amines.
Steps involved in Gabriel Phthalimide synthesis:
-The potassium or sodium salts of phthalimide are N-alkylated with the primary alkyl halide to obtain the corresponding N-alkyl phthalimide.
-The phthalimide obtained by alkylation is not a nucleophile and hence does not react further. Therefore hydrolyzation of this intermediate phthalimide is necessary.
-Further, the alkyl phthalimide obtained undergoes hydrolysis to obtain primary amine.
Gabriel Phthalimide reaction:

Note: The aryl amines cannot be prepared by Gabriel phthalimide synthesis because aryl halides do not undergo simple nucleophilic substitution. The huge benefit of Gabriel phthalimide synthesis is that it avoids over alkylation.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

The number of chromosomes in male grasshopper is a class 12 biology CBSE

Verhulst Pearl logistic growth described by the equation class 12 biology CBSE

Cry II Ab and cry I Ab produce toxins that control class 12 biology CBSE




