Write the reaction of an amino acid with ninhydrin reagent?
Answer
610.8k+ views
Hint: Amino acid are the compounds which consists of amino and the carboxylic group and when they are made to react with the laboratory reagent ninhydrin gives the purple color product (major product) along with the carbon carbon dioxide and aldehyde.
Complete step by step answer:
Amino acids are the organic compounds containing both the amino group and carboxylic group in their molecules. They are represented by the general formula as:
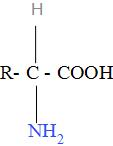
Here, -C- is called alpha carbon because the carbon atom attached next to the carboxylic group is called alpha carbon.
R is the side chain and is different for different amino acids.
-COOH is the carboxylic group.
$-\text{N}{{\text{H}}_{2}}$ is the amino group.
In the amino acid , the acidic properties are due to the $-\text{NH}_{3}^{+}$ group and the basic properties are due to the $\text{-CO}{{\text{O}}^{-}}$.
On the other hand, ninhydrin is an organic compound and is tricycle 1,2,3 -trione and is a very strong oxidizing agent ( that oxidizes the other substance).
When amino acids are made to react with the ninhydrin, they undergo decarboxylation (i.e. there is removal of carbon dioxide) and it produces carbon dioxide , ammonia and an aldehyde and the ninhydrin which had undergone reduction further reacts with the ammonia and produces purple colored complex which is known as the Ruhemann’s purple. The reaction occurs as:
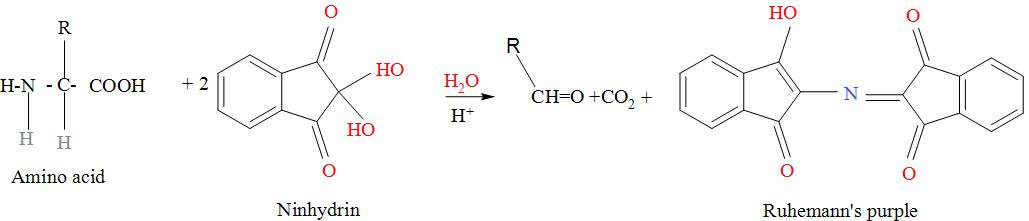
Note: To detect the presence of amino acids , they’re made to react with the ninhydrin reagent which is basically yellow in colour, but when reacted with the amino acids, the resultant product is in purple colour which indicates that amino acid is present in that very compound.
Complete step by step answer:
Amino acids are the organic compounds containing both the amino group and carboxylic group in their molecules. They are represented by the general formula as:

Here, -C- is called alpha carbon because the carbon atom attached next to the carboxylic group is called alpha carbon.
R is the side chain and is different for different amino acids.
-COOH is the carboxylic group.
$-\text{N}{{\text{H}}_{2}}$ is the amino group.
In the amino acid , the acidic properties are due to the $-\text{NH}_{3}^{+}$ group and the basic properties are due to the $\text{-CO}{{\text{O}}^{-}}$.
On the other hand, ninhydrin is an organic compound and is tricycle 1,2,3 -trione and is a very strong oxidizing agent ( that oxidizes the other substance).
When amino acids are made to react with the ninhydrin, they undergo decarboxylation (i.e. there is removal of carbon dioxide) and it produces carbon dioxide , ammonia and an aldehyde and the ninhydrin which had undergone reduction further reacts with the ammonia and produces purple colored complex which is known as the Ruhemann’s purple. The reaction occurs as:

Note: To detect the presence of amino acids , they’re made to react with the ninhydrin reagent which is basically yellow in colour, but when reacted with the amino acids, the resultant product is in purple colour which indicates that amino acid is present in that very compound.
Recently Updated Pages
Master Class 10 English: Engaging Questions & Answers for Success

Master Class 10 Social Science: Engaging Questions & Answers for Success

Master Class 10 Computer Science: Engaging Questions & Answers for Success

Class 10 Question and Answer - Your Ultimate Solutions Guide

Master Class 10 General Knowledge: Engaging Questions & Answers for Success

Master Class 10 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Ribosomal RNA is actively synthesised in A Nucleoplasm class 12 biology CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE




