Write the IUPAC name of the compound in the picture.
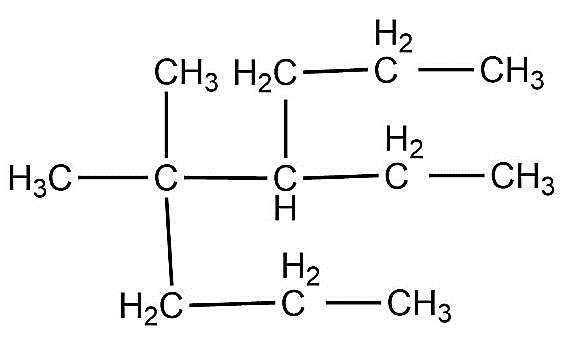

Answer
618.3k+ views
Hint: Find out the longest chain present in the compound. Determine whether there are any functional groups or substituents. Accordingly, assign priority numbers and name the compound.
Complete step by step answer:
Let’s have a look at the question,
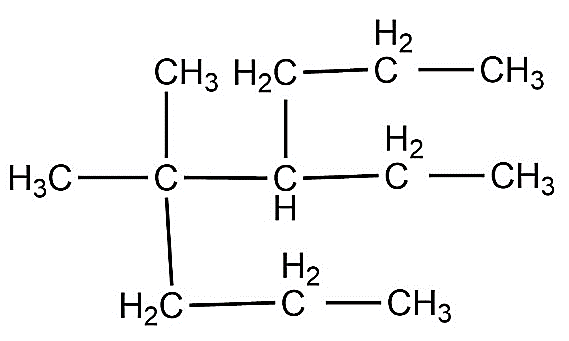
-According to IUPAC rules, we need to first determine the longest hydrocarbon chain present in the compound i.e. hydrocarbon backbone of the molecule. Here we can see that our longest carbon chain consists of eight carbon atoms and the molecule doesn’t have any unsaturation in it, so it is an octane.
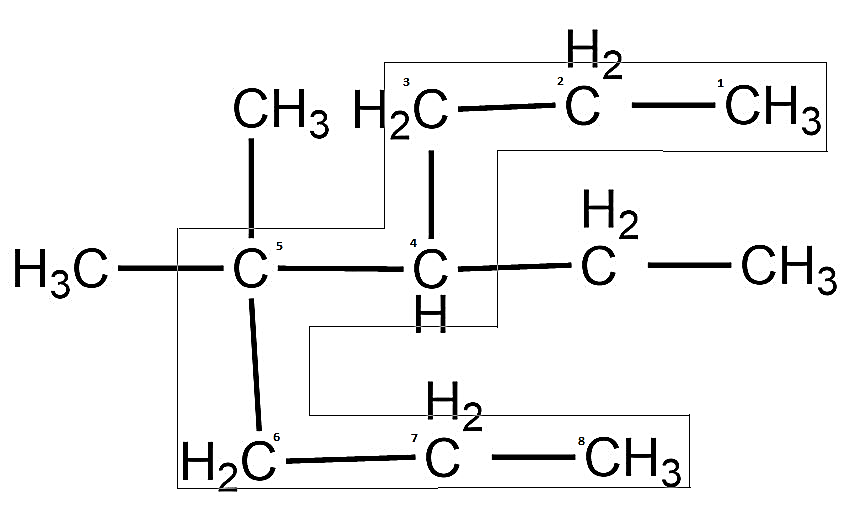
-After we determine the longest carbon chain, next we need to start numbering, so can see that there are two substituents in this molecule. One is ethyl and there are two methyl groups.
-While assigning the number, the substituent having a higher number of carbon atoms in the chain gets higher priority. That means, ethyl has greater priority than methyl group.
-So, when we keep in mind the priority rules, a carbon atom having two methyl groups gets the number 5 and the one adjacent to it having an ethyl group gets the number 4.
-Therefore, it now has an ethyl group at fourth position so it becomes 4-ethyl and two methyl groups are present at fifth position so it is written as 5, 5-dimethyl.
-Now, once we have assigned names to all the species present in the compound, we just need to put all the names together. So, the IUPAC name of a given compound is 4-ethyl-5, 5-dimethyloctane.
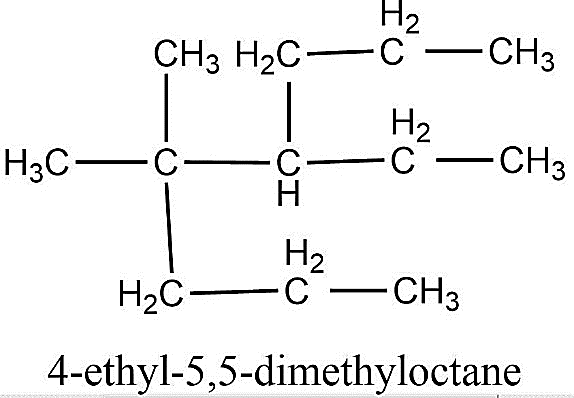
The name of the given compound is 4-ethyl-5,5-dimethyloctane.
Note: Remember that the substituents are arranged in alphabetical order in the IUPAC name. Also, note that di-, tri-,tetra- is not considered while arranging them in alphabetical order and so here even though dimethyl comes before ethyl alphabetically, ethyl is written first because ethyl comes before methyl in alphabetical order.
Complete step by step answer:
Let’s have a look at the question,

-According to IUPAC rules, we need to first determine the longest hydrocarbon chain present in the compound i.e. hydrocarbon backbone of the molecule. Here we can see that our longest carbon chain consists of eight carbon atoms and the molecule doesn’t have any unsaturation in it, so it is an octane.

-After we determine the longest carbon chain, next we need to start numbering, so can see that there are two substituents in this molecule. One is ethyl and there are two methyl groups.
-While assigning the number, the substituent having a higher number of carbon atoms in the chain gets higher priority. That means, ethyl has greater priority than methyl group.
-So, when we keep in mind the priority rules, a carbon atom having two methyl groups gets the number 5 and the one adjacent to it having an ethyl group gets the number 4.
-Therefore, it now has an ethyl group at fourth position so it becomes 4-ethyl and two methyl groups are present at fifth position so it is written as 5, 5-dimethyl.
-Now, once we have assigned names to all the species present in the compound, we just need to put all the names together. So, the IUPAC name of a given compound is 4-ethyl-5, 5-dimethyloctane.

The name of the given compound is 4-ethyl-5,5-dimethyloctane.
Note: Remember that the substituents are arranged in alphabetical order in the IUPAC name. Also, note that di-, tri-,tetra- is not considered while arranging them in alphabetical order and so here even though dimethyl comes before ethyl alphabetically, ethyl is written first because ethyl comes before methyl in alphabetical order.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




