The structure of $S{{O}_{3}}$ molecule in the gaseous phase contains:
(A) Only $\sigma $- bond between sulphur and oxygen
(B) $\sigma $ bonds and a (p$\pi $-p$\pi $) bonds between sulphur and oxygen
(C) $\sigma $bonds and a d$\pi $- p$\pi $) bonds between sulphur and oxygen
(D) $\sigma $ bonds and a (p$\pi $-p$\pi $) bonds and a ( d$\pi $- p$\pi $) bonds between sulphur and oxygen
Answer
597k+ views
Hint: To answer this question we should be aware when $\sigma $bonds, (p$\pi $-p$\pi $) bonds and ( d$\pi $- p$\pi $) bonds are formed. Valence electrons are the electrons that are present in the outermost shell of an atom upon this the covalency of the atom depends.
Complete answer:
Sulphur has six electrons in its outermost shell that is its covalency is six. Covalency of an atom is the number bonds an atom can make. In solid state the $S{{O}_{3}}$molecule exists in a cluster that is the number of$S{{O}_{3}}$ molecules bonded together to form a solid structure. In the gas phase the $S{{O}_{3}}$ molecule exists as a single molecule which is also said to exist in the free State.
$\sigma $- bond is formed by overlapping of atomic orbitals or hybrid orbitals in their axis.
For example: overlap of s-s, s-p,$s{{p}^{2}}$-$s{{p}^{2}}$ overlap along their axis.
\[\pi \]- bond is formed by the side to side overlap of molecular orbital or atomic orbital along a plane perpendicular to the plane. If the donating orbital is p and the orbital to which the electron pair is donated is p then it is known as P\[\pi \]-p\[\pi \].
Similarly, if the donating orbital is d and the orbital to which the electron pair is donated is p then it is known as d$\pi $- p$\pi $
Firstly, let's draw the Lewis structure of$S{{O}_{3}}$:
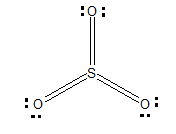
There are three delocalised \[\pi \] bonds, out of which one of them is P\[\pi \]-p\[\pi \]and other two are d$\pi $- p$\pi $. The sulphur is $d\pi -p\pi $hybridized.
The electronic configuration of sulphur is $1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{4}}$.
Sulphur in ground state:
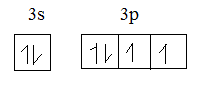
In excited state:
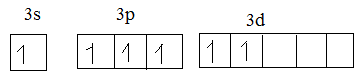
Oxygen has 6 electrons. So, the valence electrons of oxygen make bond with unpaired electrons.
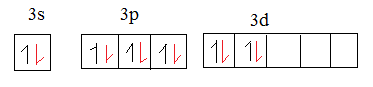
The 6 valence electrons in oxygen are of 2p orbital.
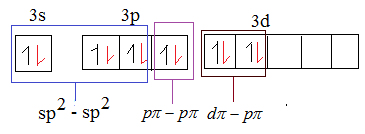
The red coloured electrons in the above mentioned diagram are the electrons of oxygen. The $s{{p}^{2}}$-$s{{p}^{2}}$ overlap forms $\sigma $ bond as sulphur in $S{{O}_{3}}$ is $s{{p}^{2}}$ hybridized.
From the above diagram it is evident that option D is the correct answer.
Note: Knowing the valance electrons of a particular element may help us to figure out their structure. Always whenever, a double bond is formed. Here one of the bonds will be $\sigma $ bond and the other will be $\pi $ bond. Now, the $\pi $ bond can be p\[\pi \]-p\[\pi \]bond, d$\pi $- p$\pi $bond or d\[\pi \]-d\[\pi \]bond.
Complete answer:
Sulphur has six electrons in its outermost shell that is its covalency is six. Covalency of an atom is the number bonds an atom can make. In solid state the $S{{O}_{3}}$molecule exists in a cluster that is the number of$S{{O}_{3}}$ molecules bonded together to form a solid structure. In the gas phase the $S{{O}_{3}}$ molecule exists as a single molecule which is also said to exist in the free State.
$\sigma $- bond is formed by overlapping of atomic orbitals or hybrid orbitals in their axis.
For example: overlap of s-s, s-p,$s{{p}^{2}}$-$s{{p}^{2}}$ overlap along their axis.
\[\pi \]- bond is formed by the side to side overlap of molecular orbital or atomic orbital along a plane perpendicular to the plane. If the donating orbital is p and the orbital to which the electron pair is donated is p then it is known as P\[\pi \]-p\[\pi \].
Similarly, if the donating orbital is d and the orbital to which the electron pair is donated is p then it is known as d$\pi $- p$\pi $
Firstly, let's draw the Lewis structure of$S{{O}_{3}}$:

There are three delocalised \[\pi \] bonds, out of which one of them is P\[\pi \]-p\[\pi \]and other two are d$\pi $- p$\pi $. The sulphur is $d\pi -p\pi $hybridized.
The electronic configuration of sulphur is $1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{4}}$.
Sulphur in ground state:

In excited state:

Oxygen has 6 electrons. So, the valence electrons of oxygen make bond with unpaired electrons.

The 6 valence electrons in oxygen are of 2p orbital.

The red coloured electrons in the above mentioned diagram are the electrons of oxygen. The $s{{p}^{2}}$-$s{{p}^{2}}$ overlap forms $\sigma $ bond as sulphur in $S{{O}_{3}}$ is $s{{p}^{2}}$ hybridized.
From the above diagram it is evident that option D is the correct answer.
Note: Knowing the valance electrons of a particular element may help us to figure out their structure. Always whenever, a double bond is formed. Here one of the bonds will be $\sigma $ bond and the other will be $\pi $ bond. Now, the $\pi $ bond can be p\[\pi \]-p\[\pi \]bond, d$\pi $- p$\pi $bond or d\[\pi \]-d\[\pi \]bond.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




