The reaction of PhOH with aldehyde RCHO in acidic medium gives:
Answer
542.1k+ views
Hint: We know that phenol is an aromatic organic compound with the molecular formula. In biochemistry, an aldehyde is a major functional group with the general formula RCHO. This formula consists of a carbonyl functional group, which uses the carbon's two remaining bonds to bind to hydrogen, and an R functional group, which may be second hydrogen, an alkyl, or an aryl group.
Complete answer:
We know that, PhOH is ortho directing and para directing. So, in acidic medium, the aldehyde RCHO is bound to ortho and para positions. Because of steric hindrance, the ortho product is less durable, and the para product is the most common.
This reaction can be represented as:
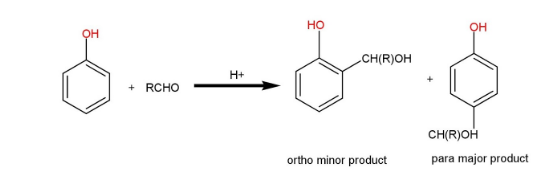
We can define this reaction as, under neutral conditions alcohols are relatively weaker nucleophiles and bind steadily to the group. However, in an acidic medium, the velocity increases as the group protonates, raising the positive charge on the carbonyl C atom and making it more reactive. Since \[R{O^ - }\] is in equilibrium with OH in simple medium and reacts with the group faster than ROH. Furthermore, \[R{O^ - }\] is a strong nucleophile.
Hence, we can conclude that, para product is the most common in this reaction.
Note:
We should remember that the electron density in ortho is lower than in para. As a result, ortho is a minor commodity and para is a main product. Also, the para compound is usually the main product when an ortho-para directing substituent is present on the benzene ring for an electrophilic aromatic substitution reaction.
Complete answer:
We know that, PhOH is ortho directing and para directing. So, in acidic medium, the aldehyde RCHO is bound to ortho and para positions. Because of steric hindrance, the ortho product is less durable, and the para product is the most common.
This reaction can be represented as:

We can define this reaction as, under neutral conditions alcohols are relatively weaker nucleophiles and bind steadily to the group. However, in an acidic medium, the velocity increases as the group protonates, raising the positive charge on the carbonyl C atom and making it more reactive. Since \[R{O^ - }\] is in equilibrium with OH in simple medium and reacts with the group faster than ROH. Furthermore, \[R{O^ - }\] is a strong nucleophile.
Hence, we can conclude that, para product is the most common in this reaction.
Note:
We should remember that the electron density in ortho is lower than in para. As a result, ortho is a minor commodity and para is a main product. Also, the para compound is usually the main product when an ortho-para directing substituent is present on the benzene ring for an electrophilic aromatic substitution reaction.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




