The oxidation number of Co in the complex ion is:
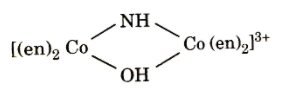
(A) +3
(B) +2
(C) +4
(D) +6

Answer
555.1k+ views
Hint: To solve this problem we have to consider the oxidation state of cobalt as x and then by adding the oxidation state of the other elements which will be equal to the net charge present in the molecule.
Complete step by step solution:
In the given question we have to calculate the oxidation state of Co in the given complex. So firstly we should know that the given complex is a coordination compound in which the molecules are attached are an en group which is also known as ethylenediamine a chlorine molecule.
Ethylenediamine is a type of bidentate ligand which means that this molecule consists of two donor atoms but it is a neutral atom which means that the oxidation state is 0.
Let the oxidation state of cobalt is x and we know that the oxidation state of NH is -2 and OH is -1 and the total charge is +3 hence the oxidation state of Co is :
\[\begin{align}
& 2(x)+1(-2)+1(-1)=+3 \\
& x=+3 \\
\end{align}\]
Hence, the correct answer is option (A) i.e. the oxidation number of Co in the complex ion is +3.
Note: In the given molecule the ligand which has two donor sites is known as a bidentate ligand. When a ligand has multiple sites which act as a donor atom then it forms the ring-like structure called chelate and the ligand is known as the chelating ligand which forms a stable structure. Oxidation state is the number which is assigned to an element in chemical combination which usually represents the number of electrons which are loosed or gained by that element.
Complete step by step solution:
In the given question we have to calculate the oxidation state of Co in the given complex. So firstly we should know that the given complex is a coordination compound in which the molecules are attached are an en group which is also known as ethylenediamine a chlorine molecule.
Ethylenediamine is a type of bidentate ligand which means that this molecule consists of two donor atoms but it is a neutral atom which means that the oxidation state is 0.
Let the oxidation state of cobalt is x and we know that the oxidation state of NH is -2 and OH is -1 and the total charge is +3 hence the oxidation state of Co is :
\[\begin{align}
& 2(x)+1(-2)+1(-1)=+3 \\
& x=+3 \\
\end{align}\]
Hence, the correct answer is option (A) i.e. the oxidation number of Co in the complex ion is +3.
Note: In the given molecule the ligand which has two donor sites is known as a bidentate ligand. When a ligand has multiple sites which act as a donor atom then it forms the ring-like structure called chelate and the ligand is known as the chelating ligand which forms a stable structure. Oxidation state is the number which is assigned to an element in chemical combination which usually represents the number of electrons which are loosed or gained by that element.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




