The number of neighbours around each particle in a face-centred cubic lattice is:
A.4
B.6
C.12
D.8
Answer
622.5k+ views
Hint:
In a face centred cubic (fcc) lattice, the atoms of the given substance are located on all 6 sides of the cubic structure as well as on all the 8 vertices of the cube.
Complete step by step answer:
The geometrical representation or the structure of a face centred cubic lattice can be visually represented as follows:
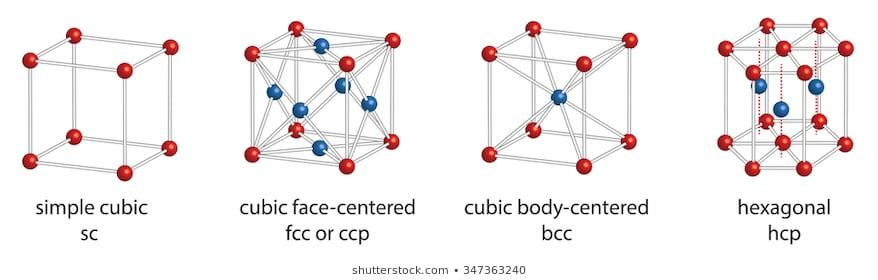
As the name suggests, a Face Centred Cubic (fcc) lattice structure has atoms on all the sides of the cubic structure as well as all the vertices. There is no atom present in the centre of the cube. This brings the total count of the total number of atoms present in a Face Centred Cubic structure to 12. Considering that each vertex contains \[{\dfrac{1}{8}^{th}}\]of an atom and each face contains half an atom, the total number of atoms present in face centred cubic lattice is :
= \[\dfrac{1}{8}\](number of vertices) + \[\dfrac{1}{2}\](number of sides)
= \[\dfrac{1}{8}\](8) + \[\dfrac{1}{2}\](6)
\[ = {\text{ }}1{\text{ }} + {\text{ }}3\]
\[ = {\text{ }}4 \]atoms
Hence in FCC, there are 4 atoms per unit cell.
-In order to understand the number of neighbours around each particle in a face-centred cubic, we must understand a concept known as Coordination number.
-In chemistry, crystallography, and materials science, the coordination number, also called ligancy, of a central atom in a molecule or crystal is the number of atoms, molecules or ions bonded to it.
-The coordination number is fixed for a certain type of lattice structure. And for Face Centred Cubic lattice structure, the coordination number is 12.
-Hence, by definition, the number of neighbours around each particle in a face-centred cubic lattice is 12.
Hence, Option C is the correct option.
Note:
The coordination number for the various types of cubic arrangements are as follows:
Simple cubic lattice: 6
Body centred cubic lattice: 8
Face centred cubic lattice: 12
In a face centred cubic (fcc) lattice, the atoms of the given substance are located on all 6 sides of the cubic structure as well as on all the 8 vertices of the cube.
Complete step by step answer:
The geometrical representation or the structure of a face centred cubic lattice can be visually represented as follows:

As the name suggests, a Face Centred Cubic (fcc) lattice structure has atoms on all the sides of the cubic structure as well as all the vertices. There is no atom present in the centre of the cube. This brings the total count of the total number of atoms present in a Face Centred Cubic structure to 12. Considering that each vertex contains \[{\dfrac{1}{8}^{th}}\]of an atom and each face contains half an atom, the total number of atoms present in face centred cubic lattice is :
= \[\dfrac{1}{8}\](number of vertices) + \[\dfrac{1}{2}\](number of sides)
= \[\dfrac{1}{8}\](8) + \[\dfrac{1}{2}\](6)
\[ = {\text{ }}1{\text{ }} + {\text{ }}3\]
\[ = {\text{ }}4 \]atoms
Hence in FCC, there are 4 atoms per unit cell.
-In order to understand the number of neighbours around each particle in a face-centred cubic, we must understand a concept known as Coordination number.
-In chemistry, crystallography, and materials science, the coordination number, also called ligancy, of a central atom in a molecule or crystal is the number of atoms, molecules or ions bonded to it.
-The coordination number is fixed for a certain type of lattice structure. And for Face Centred Cubic lattice structure, the coordination number is 12.
-Hence, by definition, the number of neighbours around each particle in a face-centred cubic lattice is 12.
Hence, Option C is the correct option.
Note:
The coordination number for the various types of cubic arrangements are as follows:
Simple cubic lattice: 6
Body centred cubic lattice: 8
Face centred cubic lattice: 12
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 Chemistry: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Name the crygenes that control cotton bollworm and class 12 biology CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

How many molecules of ATP and NADPH are required information class 12 biology CBSE

In a transcription unit the promoter is said to be class 12 biology CBSE




