The monomer of neoprene is:
A.1,3-Butadiene
B.2-Chloro-1,3-butadiene
C.2-Methyl-1,3-butadiene
D.vinyl chloride
Answer
609k+ views
Hint: It is an Addition homopolymer that exhibits rubber-like properties. It takes place through free radical polymerization. Identify the polymer units of given monomer units. The monomer unit of neoprene is also known as chloroprene. Then, the monomer of neoprene could be known.
Complete step by step answer:
-First, we will discuss polymerization. When the monomer units combine together to form polymer chains in a chemical reaction, it is termed as polymerization.
-Now, we are given the four monomer units. Let us discuss it one by one.
1.The first is 1,3–Butadiene. It is copolymerized with other monomers like styrene, and the most commonly formed copolymer is styrene-butadiene.
2.The second is 2-Chloro-1,3-butadiene. It is also known as chloroprene, and it is the monomer unit of neoprene.
The structure of monomer 2-chloro-1-3butadiene is given below. It is also called chloroprene.
The reaction that undergoes is given below for better understanding.
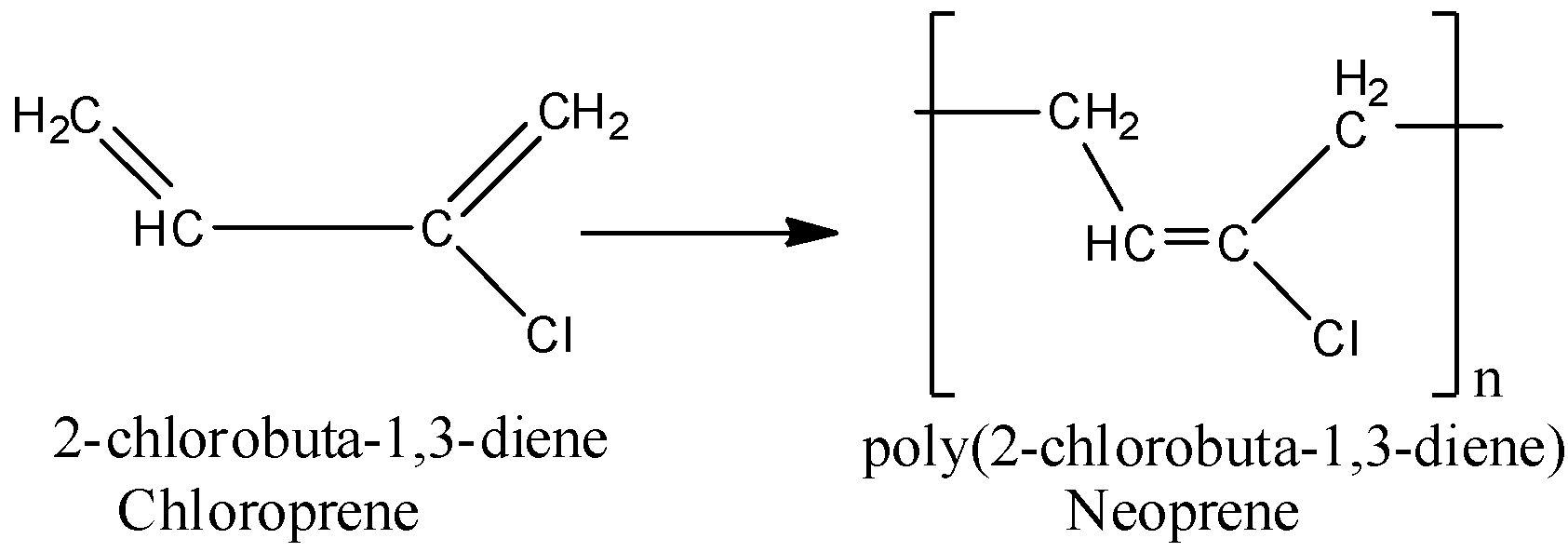
3.The third is 2-Methyl-1,3-butadiene. It is also known as isoprene, and it is the monomer unit of polyisoprene.
4.The fourth is vinyl chloride. It is also known as chloroethene, and it is the monomer unit of polyvinyl chloride.
Thus, in the last we can conclude that the monomer unit of neoprene is 2-Chloro-1,3-butadiene.
Hence, the correct option is (B).
Note:
Neoprene belongs to the family of synthetic rubbers. It exhibits the property of flexibility, and is used in a large number of applications like laptop sleeves, and electrical insulation.
Complete step by step answer:
-First, we will discuss polymerization. When the monomer units combine together to form polymer chains in a chemical reaction, it is termed as polymerization.
-Now, we are given the four monomer units. Let us discuss it one by one.
1.The first is 1,3–Butadiene. It is copolymerized with other monomers like styrene, and the most commonly formed copolymer is styrene-butadiene.
2.The second is 2-Chloro-1,3-butadiene. It is also known as chloroprene, and it is the monomer unit of neoprene.
The structure of monomer 2-chloro-1-3butadiene is given below. It is also called chloroprene.

The reaction that undergoes is given below for better understanding.

3.The third is 2-Methyl-1,3-butadiene. It is also known as isoprene, and it is the monomer unit of polyisoprene.
4.The fourth is vinyl chloride. It is also known as chloroethene, and it is the monomer unit of polyvinyl chloride.
Thus, in the last we can conclude that the monomer unit of neoprene is 2-Chloro-1,3-butadiene.
Hence, the correct option is (B).
Note:
Neoprene belongs to the family of synthetic rubbers. It exhibits the property of flexibility, and is used in a large number of applications like laptop sleeves, and electrical insulation.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




