The correct electron dot structure of water molecule is :
(A)

(B)
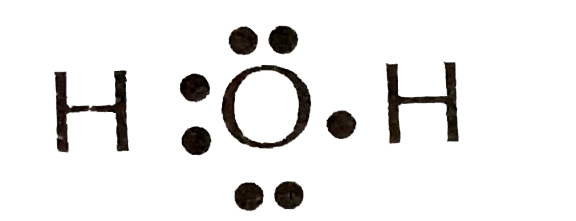
(C)

(D)

Answer
565.9k+ views
Hint: The atomic number of oxygen is 8. So, we can say that there are 6 valence electrons. And the atomic number of hydrogen is 1. So, the number of valence electrons is 1.
Complete step by step answer:
The electronic configuration of oxygen is 2, 6. So there are 6 valence electrons available for oxygen in order to form a chemical bonding. Similarly, the electronic configuration of hydrogen is 1. So, there is 1 valence electron available for hydrogen in order to form a chemical bonding.
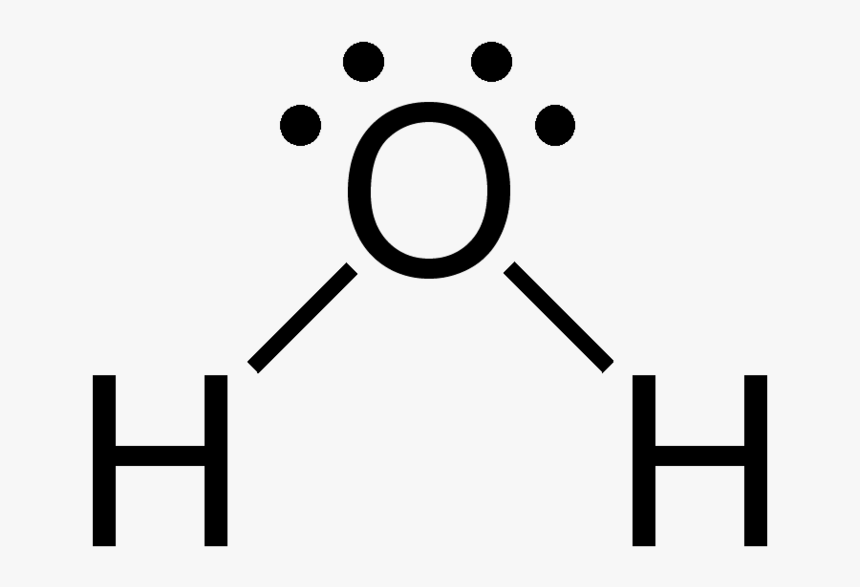
In the diagram, provided above we get an idea about the structure of water molecules.
Oxygen atom has 6 valence electrons out of which 2 valence electrons are involved in bonding with the 2 hydrogen atoms. Therefore, a water molecule has 2 bond pairs of electrons and 2 lone pair (non-bonded) pairs of electrons.
So, we can say that the Lewis dot structure of of water molecule is:

Therefore, corresponding to the question, we can that the correct Lewis dot structure of water molecules is number 3.
So, Option C is the correct answer.
Note:
(1) Lewis structures also known as Lewis dot structures or electron dot structures are diagrams that represent the valence electrons of an atom within a molecule. These Lewis symbols are Lewis structures which help visualize the valence electrons of atoms and molecules, whether they exist as lone pairs or within bonds.
(2) In order to draw the Lewis dot structure, place any remaining lone electron pairs on the central atom. Usually, electronegative atoms will have lone pairs on them. The number of electrons placed on the molecule should correspond to the total number of valence electrons of the atoms in the molecule.
Complete step by step answer:
The electronic configuration of oxygen is 2, 6. So there are 6 valence electrons available for oxygen in order to form a chemical bonding. Similarly, the electronic configuration of hydrogen is 1. So, there is 1 valence electron available for hydrogen in order to form a chemical bonding.

In the diagram, provided above we get an idea about the structure of water molecules.
Oxygen atom has 6 valence electrons out of which 2 valence electrons are involved in bonding with the 2 hydrogen atoms. Therefore, a water molecule has 2 bond pairs of electrons and 2 lone pair (non-bonded) pairs of electrons.
So, we can say that the Lewis dot structure of of water molecule is:

Therefore, corresponding to the question, we can that the correct Lewis dot structure of water molecules is number 3.
So, Option C is the correct answer.
Note:
(1) Lewis structures also known as Lewis dot structures or electron dot structures are diagrams that represent the valence electrons of an atom within a molecule. These Lewis symbols are Lewis structures which help visualize the valence electrons of atoms and molecules, whether they exist as lone pairs or within bonds.
(2) In order to draw the Lewis dot structure, place any remaining lone electron pairs on the central atom. Usually, electronegative atoms will have lone pairs on them. The number of electrons placed on the molecule should correspond to the total number of valence electrons of the atoms in the molecule.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding the Different Types of Solutions in Chemistry

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




