The compound of molecular formula ${ C }_{ 5 }{ H }_{ 10 }{ O }$(A) reacts with Tollen’s reagent to give a silver mirror but does not undergo aldol condensation. The compound A is :
A.)3-pentanone
B.)2,2-dimethyl propanal
C.)2-hydroxy-2-pentene
D.)3-methyl butanal
E.)3-methyl-2-butanone
Answer
628.8k+ views
Hint: A compound giving positive Tollens test implies that it's an aldehyde and if that compound does noy undergo aldol condensation means that the aldehyde is not having hydrogen at its $\alpha $ -position.
Complete answer:
As we mentioned in the hint, the compound giving positive Tollens test is an aldehyde. The aldehyde when introduced with Tollen’s reagent is getting oxidised into carboxylic acid.
Being a mild oxidising agent, Tollen’s reagent can’t oxidise ketone groups, so it can be used for distinguishing between aldehydes and ketones.
We can have a look at the structures of molecules in the given options.
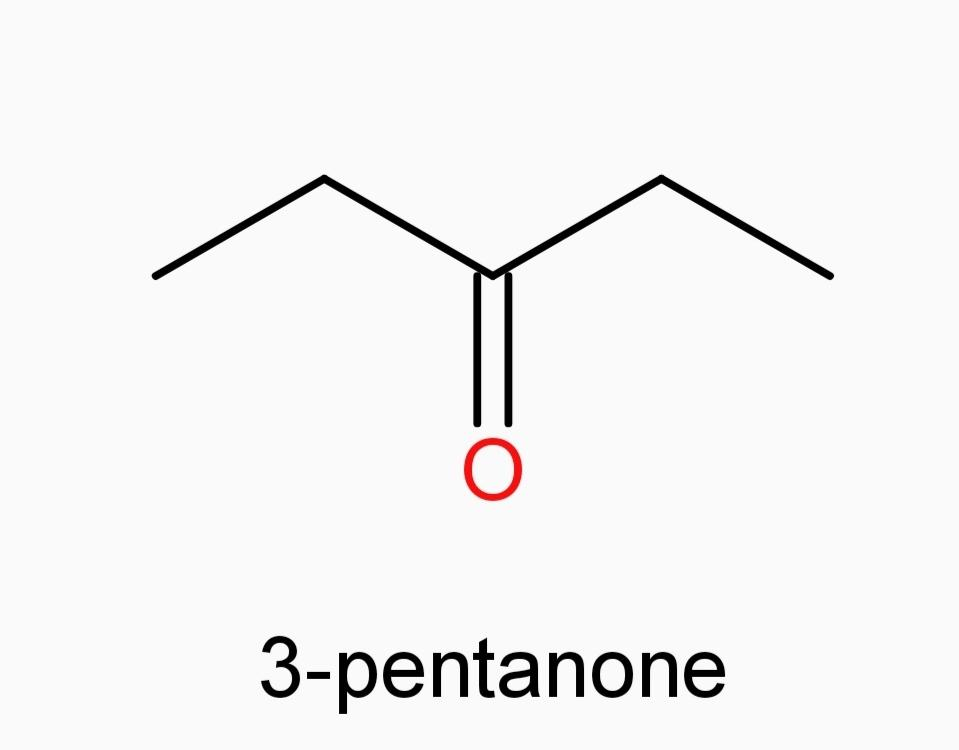
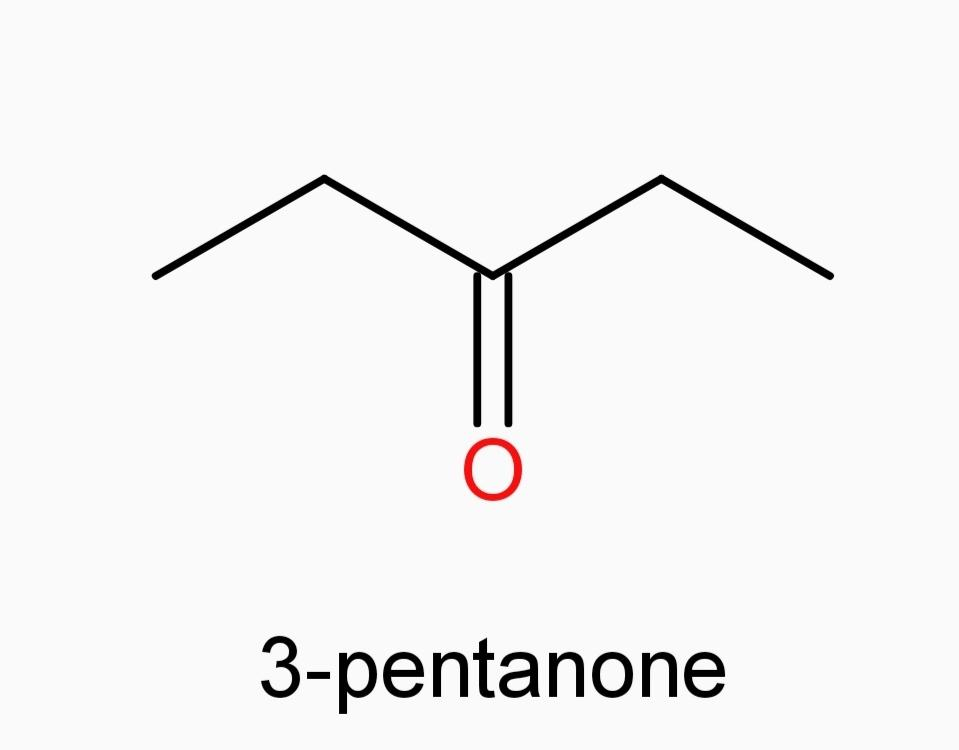
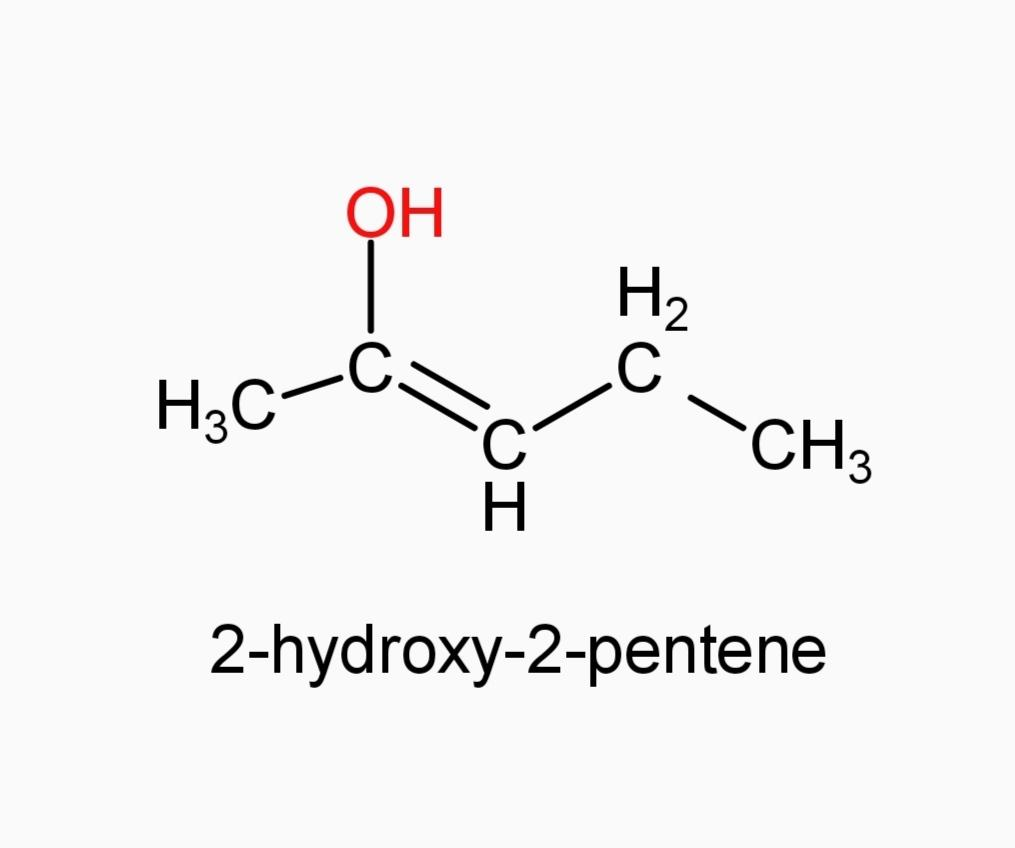
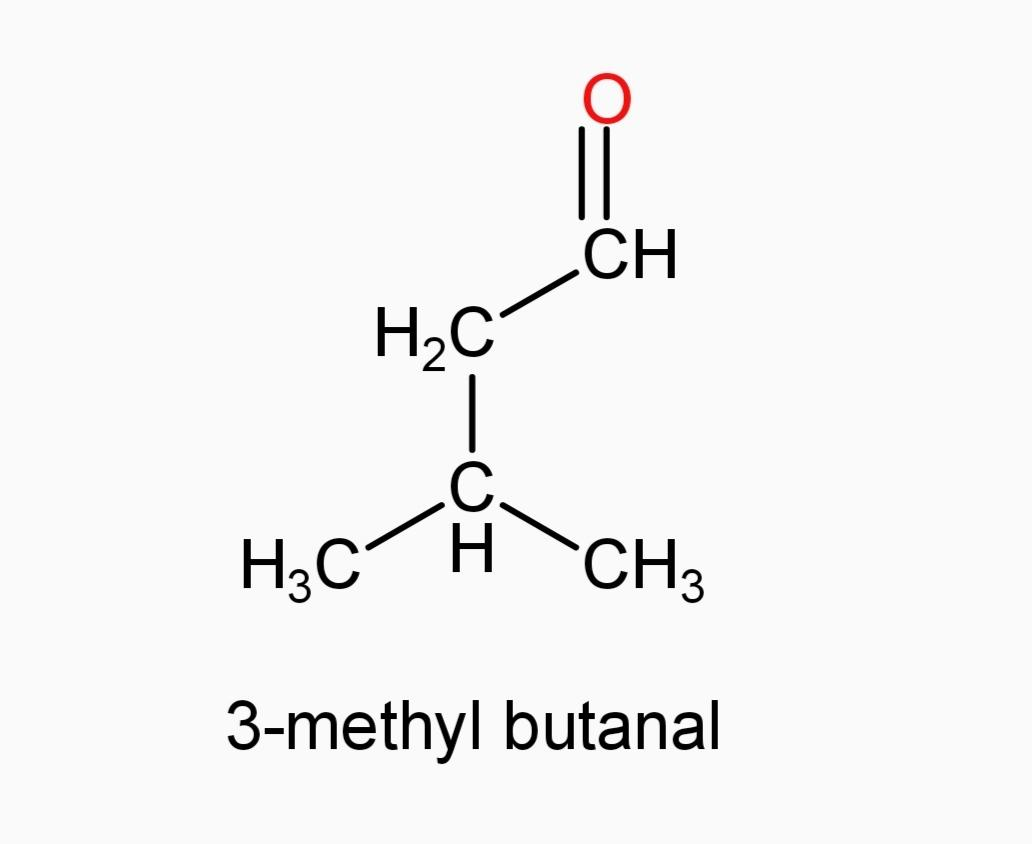
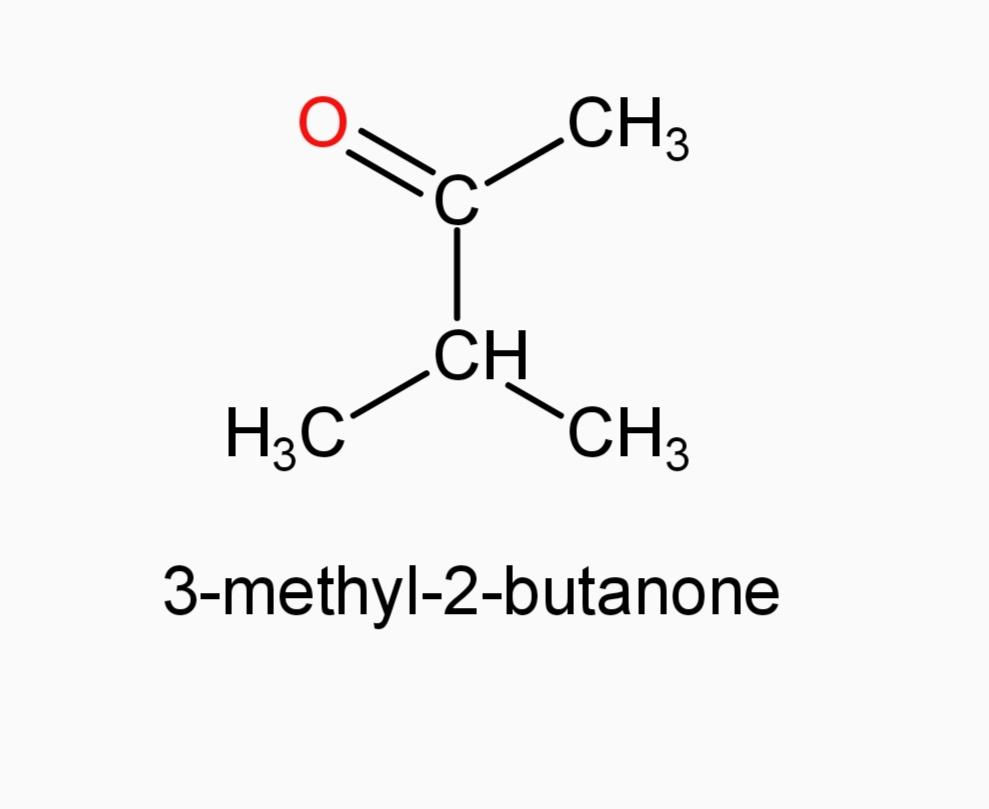
And now Option (A) 3-pentanone and Option (E) 3-methyl-2-butanone, since they are ketones, they can’t be our answer.
Along with these, Option (C) is also not the answer, since 2-hydroxy-2-pentene is an alkene and not an alcohol.
Coming to the next part of the question, i.e, the aldol condensation.
Aldol condensation occurs in aldehydes having $\alpha $-hydrogen with a dilute base to give $\beta $-hydroxy aldehydes called aldols.
If this reaction occurs between two different carbonyl compounds it is called crossed aldol condensation.
So, if a compound is giving aldol condensation, then it should have an $\alpha $-hydrogen.
Considering the rest options, i.e, Option (B)2,2-dimethyl propanal
and Option (D)3-methyl butanal
By looking at the structure of both the compounds, we can find that in the case of 3-methyl butanal, $\alpha $-hydrogen is present and so it can undergo aldol condensation.
The compound 2,2-dimethyl propanal has no $\alpha $-hydrogen present in it and therefore, it cannot undergo aldol condensation, which is in agreement with our question.
So, the correct answer is “Option B”.
Note:
Tollen’s as well as Fehling's reagent are mild oxidising agents and can be used for distinguishing between aldehydes and ketones. Tollen’s test shows some exceptions in a way that it gives positive test for formic acid, chloroform, alpha hydroxy ketones and alkyl hydroxy amines and even acetylene.
Complete answer:
As we mentioned in the hint, the compound giving positive Tollens test is an aldehyde. The aldehyde when introduced with Tollen’s reagent is getting oxidised into carboxylic acid.
Being a mild oxidising agent, Tollen’s reagent can’t oxidise ketone groups, so it can be used for distinguishing between aldehydes and ketones.
We can have a look at the structures of molecules in the given options.





And now Option (A) 3-pentanone and Option (E) 3-methyl-2-butanone, since they are ketones, they can’t be our answer.
Along with these, Option (C) is also not the answer, since 2-hydroxy-2-pentene is an alkene and not an alcohol.
Coming to the next part of the question, i.e, the aldol condensation.
Aldol condensation occurs in aldehydes having $\alpha $-hydrogen with a dilute base to give $\beta $-hydroxy aldehydes called aldols.
If this reaction occurs between two different carbonyl compounds it is called crossed aldol condensation.
So, if a compound is giving aldol condensation, then it should have an $\alpha $-hydrogen.
Considering the rest options, i.e, Option (B)2,2-dimethyl propanal
and Option (D)3-methyl butanal
By looking at the structure of both the compounds, we can find that in the case of 3-methyl butanal, $\alpha $-hydrogen is present and so it can undergo aldol condensation.
The compound 2,2-dimethyl propanal has no $\alpha $-hydrogen present in it and therefore, it cannot undergo aldol condensation, which is in agreement with our question.
So, the correct answer is “Option B”.
Note:
Tollen’s as well as Fehling's reagent are mild oxidising agents and can be used for distinguishing between aldehydes and ketones. Tollen’s test shows some exceptions in a way that it gives positive test for formic acid, chloroform, alpha hydroxy ketones and alkyl hydroxy amines and even acetylene.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Social Science: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




