
Terylene is not a:
A.Polyester fiber
B.Step growth polymer
C.Copolymer
D.Chain growth polymer.
Answer
593.4k+ views
Hint:
The polymer is large molecules also called macromolecules. They are generated from the addition or condensation of small monomers which may be monofunctional or bifunctional. Monofunctional monomers result in addition polymer while bifunctional monomers result in condensation polymer.
Complete answer:
Terylene is a type of condensation polymer generated from the condensation of ethylene glycol and terephthalic acid. Here the condensation results in the loss of water molecules. The condensation is written as:
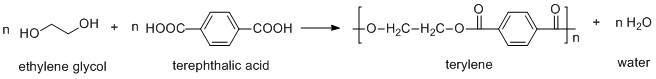
Terylene is formed by the condensation of an alcohol i.e. ethylene glycol and an acid i.e. terephthalic acid. The reaction between alcohol and acid results in the formation of an ester bond. As the monomers of ethylene glycol and terephthalic acid are linked by ester linkages and the ester is used in preparing clothes. So terylene is a polyester fiber.
Terylene is formed by condensation of repeating units of bifunctional monomers to produce long-chain polymers. In this process, the monomers combine to form dimmers then trimers then oligomers, and finally polymers. So terylene is a step-growth polymer.
Copolymers are polymers that are generated by a combination of two different units instead of a single monomer. As terylene involves two different monomer units like ethylene glycol and terephthalic acid, so it makes it a copolymer.
Chain growth polymers are polymers which are generated by the polymerization of single monomer units. They are formed by linking monomers with double and triple bonds. They are also called addition polymers. So terylene is not a chain-growth polymer as it is formed by condensation of two monomer units.
The correct answer is D, terylene is not a chain-growth polymer.
Note: The type of monomer units and the type of combination like addition or condensation has to be understood to identify the polymer. The type of polymerization is crucial to explain the properties of the polymers.
The polymer is large molecules also called macromolecules. They are generated from the addition or condensation of small monomers which may be monofunctional or bifunctional. Monofunctional monomers result in addition polymer while bifunctional monomers result in condensation polymer.
Complete answer:
Terylene is a type of condensation polymer generated from the condensation of ethylene glycol and terephthalic acid. Here the condensation results in the loss of water molecules. The condensation is written as:
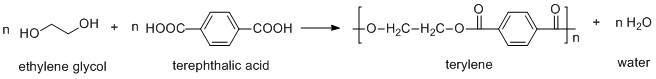
Terylene is formed by the condensation of an alcohol i.e. ethylene glycol and an acid i.e. terephthalic acid. The reaction between alcohol and acid results in the formation of an ester bond. As the monomers of ethylene glycol and terephthalic acid are linked by ester linkages and the ester is used in preparing clothes. So terylene is a polyester fiber.
Terylene is formed by condensation of repeating units of bifunctional monomers to produce long-chain polymers. In this process, the monomers combine to form dimmers then trimers then oligomers, and finally polymers. So terylene is a step-growth polymer.
Copolymers are polymers that are generated by a combination of two different units instead of a single monomer. As terylene involves two different monomer units like ethylene glycol and terephthalic acid, so it makes it a copolymer.
Chain growth polymers are polymers which are generated by the polymerization of single monomer units. They are formed by linking monomers with double and triple bonds. They are also called addition polymers. So terylene is not a chain-growth polymer as it is formed by condensation of two monomer units.
The correct answer is D, terylene is not a chain-growth polymer.
Note: The type of monomer units and the type of combination like addition or condensation has to be understood to identify the polymer. The type of polymerization is crucial to explain the properties of the polymers.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




