One mole of stachyose on hydrolysis yields:
A.1 mole of glucose + 1 mole of fructose + 2 mole of galactose
B.2 mole of glucose + 1 mole of fructose + 1 mole of galactose
C.1 mole of glucose + 2 mole of fructose + 1 mole of galactose
D.2 mole of glucose + 2 mole of fructose
Answer
624.9k+ views
Hint:
A tetrasaccharide is a saccharide or carbohydrate which upon hydrolysis gives four molecules of the same monosaccharide or different monosaccharides. The general formula of tetrasaccharides is ${{\text{C}}_{{\text{24}}}}{{\text{H}}_{{\text{42}}}}{{\text{O}}_{{\text{21}}}}$ . The monosaccharides are the simplest carbohydrates which cannot be hydrolyzed further to smaller molecules. Their general formula is ${\left( {{\text{C}}{{\text{H}}_{\text{2}}}{\text{O}}} \right)_{\text{n}}}$ where n is equal to 3-7.
Complete step by step answer:
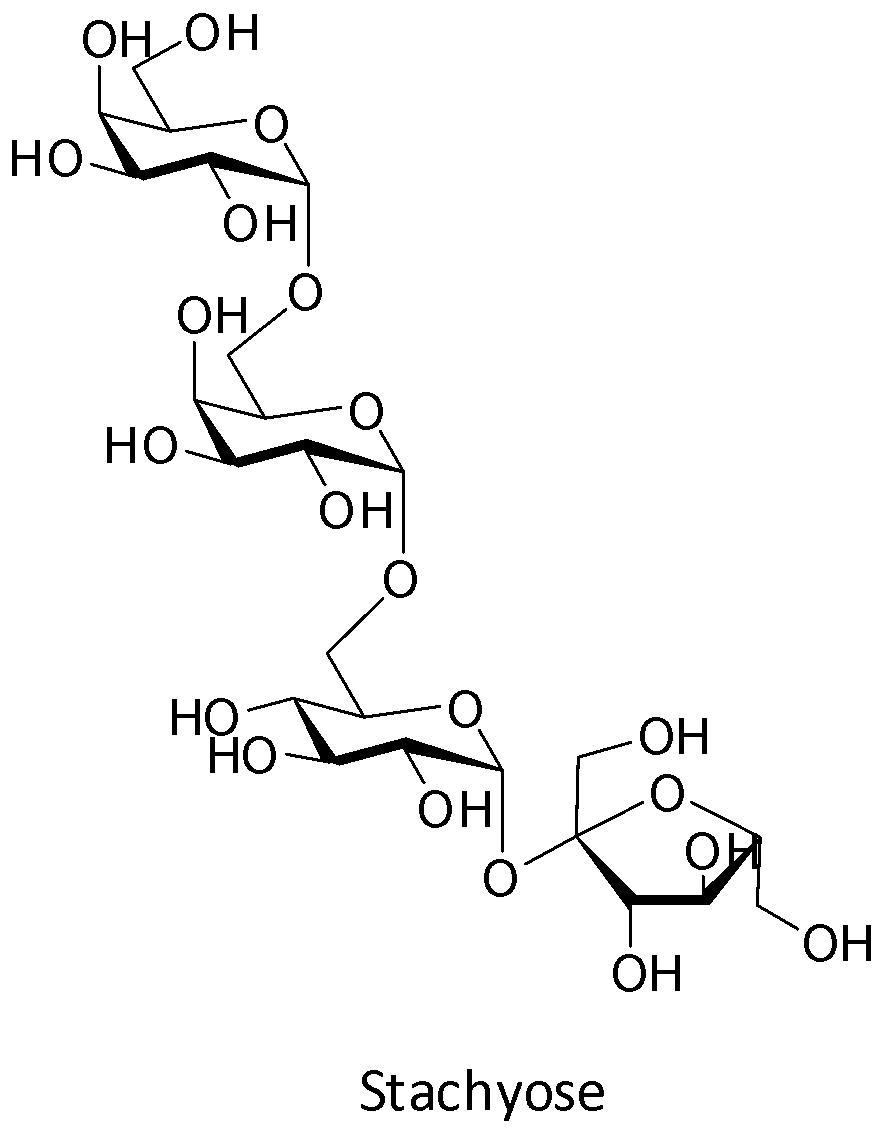
Stachyose is an example of a tetrasaccharide. It gives four molecules of different monosaccharides on complete hydrolysis. These four molecules are one molecule of glucose, one molecule of fructose and two molecules of galactose. Thus, stachyose consists of two ${\text{alpha }}$ - D - galactose units, one ${\text{alpha }}$ - D - glucose unit and one ${\text{beta }}$ - D - fructose unit. Thus, option A is the correct answer. The structure of stachyose is shown below.
The hydrolysis of a polysaccharide results in the breakage of the glycosidic linkage formed between two monosaccharide monomer units when the polysaccharide is formed. The hydrolysis of the oligosaccharide sucrose gives glucose and fructose.
The other options are not correct as there are no particular tetrasaccharides known to give any of the three combinations which are 2 mole of glucose, 1 mole of fructose, 1 mole of galactose or 1 mole of glucose, 2 mole of fructose, 1 mole of galactose or 2 mole of glucose, 2 mole of fructose. Also, the hydrolysis of stachyose does not result in these monosaccharides. Hence, the options B, C, D are not correct.
Hence option A is correct.
Note:
The carbohydrates which upon hydrolysis give two molecules of the same or different monosaccharides are called disaccharides. For example, sucrose gives glucose and fructose, maltose gives 2 molecules of glucose and lactose gives glucose and galactose. Similarly, trisaccharides give 3 molecules of the same or different monosaccharides. For example, raffinose gives one molecule each of glucose, fructose and galactose on hydrolysis.
A tetrasaccharide is a saccharide or carbohydrate which upon hydrolysis gives four molecules of the same monosaccharide or different monosaccharides. The general formula of tetrasaccharides is ${{\text{C}}_{{\text{24}}}}{{\text{H}}_{{\text{42}}}}{{\text{O}}_{{\text{21}}}}$ . The monosaccharides are the simplest carbohydrates which cannot be hydrolyzed further to smaller molecules. Their general formula is ${\left( {{\text{C}}{{\text{H}}_{\text{2}}}{\text{O}}} \right)_{\text{n}}}$ where n is equal to 3-7.
Complete step by step answer:
Stachyose is an example of a tetrasaccharide. It gives four molecules of different monosaccharides on complete hydrolysis. These four molecules are one molecule of glucose, one molecule of fructose and two molecules of galactose. Thus, stachyose consists of two ${\text{alpha }}$ - D - galactose units, one ${\text{alpha }}$ - D - glucose unit and one ${\text{beta }}$ - D - fructose unit. Thus, option A is the correct answer. The structure of stachyose is shown below.

The hydrolysis of a polysaccharide results in the breakage of the glycosidic linkage formed between two monosaccharide monomer units when the polysaccharide is formed. The hydrolysis of the oligosaccharide sucrose gives glucose and fructose.
The other options are not correct as there are no particular tetrasaccharides known to give any of the three combinations which are 2 mole of glucose, 1 mole of fructose, 1 mole of galactose or 1 mole of glucose, 2 mole of fructose, 1 mole of galactose or 2 mole of glucose, 2 mole of fructose. Also, the hydrolysis of stachyose does not result in these monosaccharides. Hence, the options B, C, D are not correct.
Hence option A is correct.
Note:
The carbohydrates which upon hydrolysis give two molecules of the same or different monosaccharides are called disaccharides. For example, sucrose gives glucose and fructose, maltose gives 2 molecules of glucose and lactose gives glucose and galactose. Similarly, trisaccharides give 3 molecules of the same or different monosaccharides. For example, raffinose gives one molecule each of glucose, fructose and galactose on hydrolysis.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




