How is Nylon 6,6 obtained. Give reaction
Answer
623.1k+ views
Hint:Nylon 6,6 is a type of polyamide or nylon. It is made up of two monomers each containing 6 carbon atoms, hexamethylenediamine and adipic acid.
Complete step by step answer:
Nylon,6,6 is synthesized by polycondensation of hexamethylenediamine and adipic acid.
Equivalent amounts of hexamethylenediamine and adipic acid are combined with water in a reactor. This is crystallized to make nylon salt, an ammonium/ carbonate mixture and thus molten nylon,6,6 is formed.
To prepare Nylon 6,6 adipic acid and hexamethylenediamine are heated at 553 K under pressure.
This reaction is a shown:
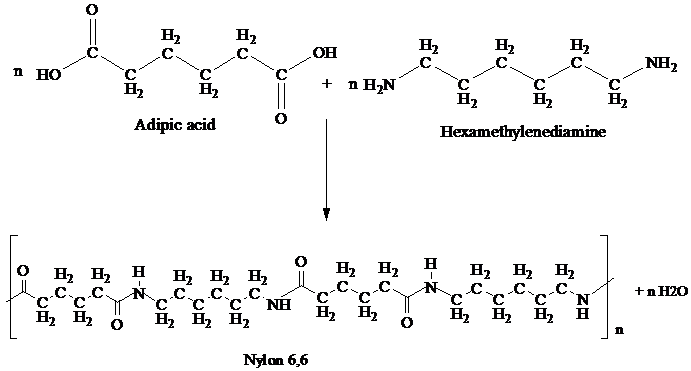
The resulting polymer is extruded into a wide range of fiber types. The fibers are drawn, or sketched in a process that increases their length and reorients the materials molecules parallel to one another to produce a strong, elastic filament.
Note:
Nylon 6,6 has long molecular chains resulting in more hydrogen bonds, creating chemical springs and making it very resilient. It is an amorphous solid so it has a large elastic property and is slightly soluble in boiling water and is very stable in nature. It is very difficult to dye, but one it is dyed it has a high colorfastness and is less susceptible to fading.
Complete step by step answer:
Nylon,6,6 is synthesized by polycondensation of hexamethylenediamine and adipic acid.
Equivalent amounts of hexamethylenediamine and adipic acid are combined with water in a reactor. This is crystallized to make nylon salt, an ammonium/ carbonate mixture and thus molten nylon,6,6 is formed.
To prepare Nylon 6,6 adipic acid and hexamethylenediamine are heated at 553 K under pressure.
This reaction is a shown:
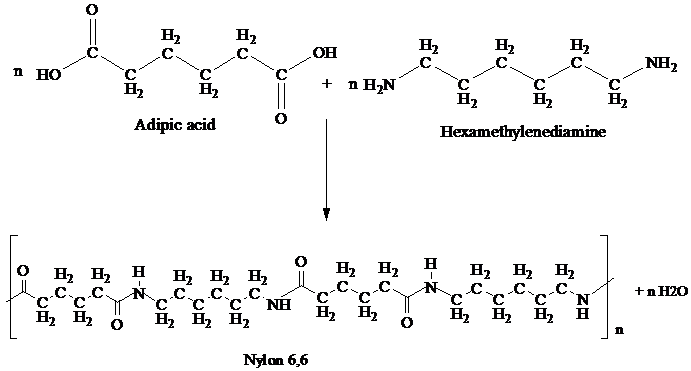
The resulting polymer is extruded into a wide range of fiber types. The fibers are drawn, or sketched in a process that increases their length and reorients the materials molecules parallel to one another to produce a strong, elastic filament.
Note:
Nylon 6,6 has long molecular chains resulting in more hydrogen bonds, creating chemical springs and making it very resilient. It is an amorphous solid so it has a large elastic property and is slightly soluble in boiling water and is very stable in nature. It is very difficult to dye, but one it is dyed it has a high colorfastness and is less susceptible to fading.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




