What is the IUPAC name for isophthalic acid?
A.Benzene-1,2-dicarboxylic acid
B.Benzene-1,3-dicarboxylic acid
C.Benzene-1.4-dicarboxylic acid
D.Benzene-1,5-dicarboxylic acid
Answer
591.6k+ views
Hint: Isophthalic acid is an organic compound, having the chemical formula: \[{{{C}}_{{6}}}{{{H}}_{{4}}}{{{(C}}{{{O}}_{{2}}}{{H)}}_{{2}}}\]. Isophthalic acid is an isomer of phthalic acid and terephthalic acid, all of them having two carboxylic acid groups of a benzene ring but the relative position of both the carboxylic acid groups is different in these three acids.
Complete step by step answer:
To predict the IUPAC name of isophthalic acid, let us first see what the structure of isophthalic acid is:
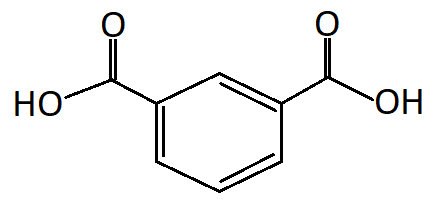
As can be seen in the above structure the two carboxylic acid groups are meta to each other or at a relative position of carbon-1 and carbon-3, the IUPAC name of isophthalic acid is Benzene-1,3-dicarboxylic acid.
Hence option B is the correct answer.
Some properties of isophthalic acid:
-Isophthalic acid is a colorless solid organic compound.
-It is mainly used for the industrial preparation of polyethylene terephthalate resin and unsaturated polyester resin.
-Isophthalate is prepared on an industrial scale by oxidizing meta-xylene using cobalt-manganese as a catalyst.
For laboratory preparations, chromic acid is used as an oxidizing agent to oxidize meta-xylene.
Every year approximately 1 billion kilograms of isophthalic acid is produced for industrial purposes.
Note:
Please note that IUPAC stands for the International Union of Pure and Applied Chemistry. IUPAC is the world authority on chemical nomenclature and terminology, including the naming of new elements in the periodic table; on standardized measurement methods, and atomic weights, and many other critically evaluated data. It is a neutral and objective scientific organization, established in 1919 by academic and industrial chemists who shared a common goal- to unite a fragmented global chemistry community for the advancement of chemical sciences via collaboration and the free exchange of scientific information.
Complete step by step answer:
To predict the IUPAC name of isophthalic acid, let us first see what the structure of isophthalic acid is:

As can be seen in the above structure the two carboxylic acid groups are meta to each other or at a relative position of carbon-1 and carbon-3, the IUPAC name of isophthalic acid is Benzene-1,3-dicarboxylic acid.
Hence option B is the correct answer.
Some properties of isophthalic acid:
-Isophthalic acid is a colorless solid organic compound.
-It is mainly used for the industrial preparation of polyethylene terephthalate resin and unsaturated polyester resin.
-Isophthalate is prepared on an industrial scale by oxidizing meta-xylene using cobalt-manganese as a catalyst.
For laboratory preparations, chromic acid is used as an oxidizing agent to oxidize meta-xylene.
Every year approximately 1 billion kilograms of isophthalic acid is produced for industrial purposes.
Note:
Please note that IUPAC stands for the International Union of Pure and Applied Chemistry. IUPAC is the world authority on chemical nomenclature and terminology, including the naming of new elements in the periodic table; on standardized measurement methods, and atomic weights, and many other critically evaluated data. It is a neutral and objective scientific organization, established in 1919 by academic and industrial chemists who shared a common goal- to unite a fragmented global chemistry community for the advancement of chemical sciences via collaboration and the free exchange of scientific information.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Which is the correct genotypic ratio of mendel dihybrid class 12 biology CBSE




