In \[PC{l_5}\], the correct statements are,
A.It has $s{p^3}d$ hybridisation with trigonal bi-pyramidal geometry.
B.It has three $Cl - P - Cl$ bond angles of ${120^0}$ and one of ${180^0}_{^{}}$.
C.Axial bond pairs suffer more repulsion from the equatorial bond pairs.
D.Equatorial $P - Cl$ bond are broken during the change $PC{l_5} \to PC{l_3} + C{l_2}$
Answer
583.5k+ views
Hint:Structure and its related information of \[PC{l_5}\] can be obtained using VSEPR Theory. In VSEPR theory, the hybridisation and structure of a molecule are predicted by counting the number of electron pairs around the central atom.
Complete step by step answer:In \[PC{l_5}\] , the central atom is Phosphorous. It has five valence electrons. We have five chlorine atoms also. Each chlorine atom contains an unpaired electron in p-orbital. The five orbitals of phosphorus containing one electron will thus undergo $s{p^3}d$ hybridisation to give five identical hybrid orbitals. Then each of this orbital will combine with the p-orbital chlorine to form \[PC{l_5}\] . There will be no lone-pair of electrons left on phosphorus. Hence the structure of \[PC{l_5}\] will be trigonal bi-pyramidal. i.e. \[PC{l_5}\] has $s{p^3}d$ hybridisation with trigonal bi-pyramidal geometry. Option A is correct.
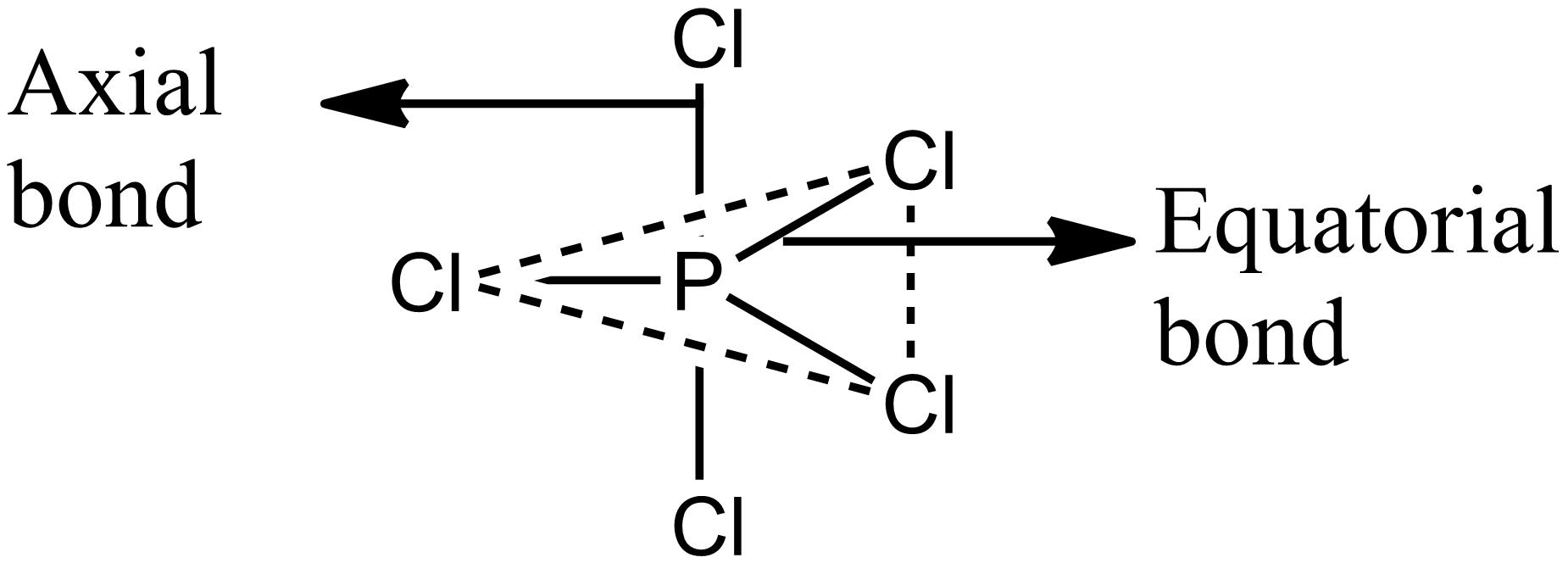
Structure of \[PC{l_5}\] is shown in the figure. It has three equatorial bonds and two axial bonds. The equatorial $Cl - P - Cl$ bond angle is ${120^0}$ and axial $Cl - P - Cl$ bond angle is ${180^0}_{^{}}$. Hence it has three $Cl - P - Cl$ bond angles of ${120^0}$ and one of ${180^0}_{^{}}$. Option B is correct.
The angle between equatorial and axial bonds is only ${90^0}$. Hence bond pair-bond pair repulsion is maximum in axial bonds compared to equatorial bonds. In order to minimize this repulsion axial bonds extend their bond length. Hence axial bonds are more elongated than equatorial bonds. Therefore the option C, axial bond pairs suffer more repulsion from the equatorial bond pairs is correct. Repulsion between equatorial bonds are negligible because their bond angle is ${120^0}$.
Since the axial bond suffers more repulsion, it is a weak bond. Hence axial bonds can be broken easily. When \[PC{l_5}\] is heated the axial bonds break to form \[PC{l_3}\] and $C{l_2}$. Equatorial bonds do not break. Hence the option D is wrong.
Therefore, the correct options are A,B and C.
Note:
The presence of elongated axial bonds make $PC{l_5}$ reactive. Here all atoms around phosphorus are Cl. If any of the Cl atom is replaced by another atom, the bond length and bond angles will change. Reactivity of molecules will also change.
Complete step by step answer:In \[PC{l_5}\] , the central atom is Phosphorous. It has five valence electrons. We have five chlorine atoms also. Each chlorine atom contains an unpaired electron in p-orbital. The five orbitals of phosphorus containing one electron will thus undergo $s{p^3}d$ hybridisation to give five identical hybrid orbitals. Then each of this orbital will combine with the p-orbital chlorine to form \[PC{l_5}\] . There will be no lone-pair of electrons left on phosphorus. Hence the structure of \[PC{l_5}\] will be trigonal bi-pyramidal. i.e. \[PC{l_5}\] has $s{p^3}d$ hybridisation with trigonal bi-pyramidal geometry. Option A is correct.

Structure of \[PC{l_5}\] is shown in the figure. It has three equatorial bonds and two axial bonds. The equatorial $Cl - P - Cl$ bond angle is ${120^0}$ and axial $Cl - P - Cl$ bond angle is ${180^0}_{^{}}$. Hence it has three $Cl - P - Cl$ bond angles of ${120^0}$ and one of ${180^0}_{^{}}$. Option B is correct.
The angle between equatorial and axial bonds is only ${90^0}$. Hence bond pair-bond pair repulsion is maximum in axial bonds compared to equatorial bonds. In order to minimize this repulsion axial bonds extend their bond length. Hence axial bonds are more elongated than equatorial bonds. Therefore the option C, axial bond pairs suffer more repulsion from the equatorial bond pairs is correct. Repulsion between equatorial bonds are negligible because their bond angle is ${120^0}$.
Since the axial bond suffers more repulsion, it is a weak bond. Hence axial bonds can be broken easily. When \[PC{l_5}\] is heated the axial bonds break to form \[PC{l_3}\] and $C{l_2}$. Equatorial bonds do not break. Hence the option D is wrong.
Therefore, the correct options are A,B and C.
Note:
The presence of elongated axial bonds make $PC{l_5}$ reactive. Here all atoms around phosphorus are Cl. If any of the Cl atom is replaced by another atom, the bond length and bond angles will change. Reactivity of molecules will also change.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




