In NPN transistors, ${{10}^{10}}$ electrons enter the emitter region in ${{10}^{-6}}s$. If 2% electrons are lost in base region then collector current and current amplification factor (β) respectively are
$\begin{align}
& A.1.57mA,49 \\
& B.1.92mA,70 \\
& C.2mA,25 \\
& D.2.25mA,100 \\
\end{align}$
Answer
604.8k+ views
Hint: First of all find out the emitter current. After that find out the collector current using the formula
${{I}_{c}}=\alpha {{I}_{e}}$
$\alpha $ is given by how much current from the emitter reaches the collector. So using this $\alpha $ amplification factor can be found out.
Complete step by step answer:
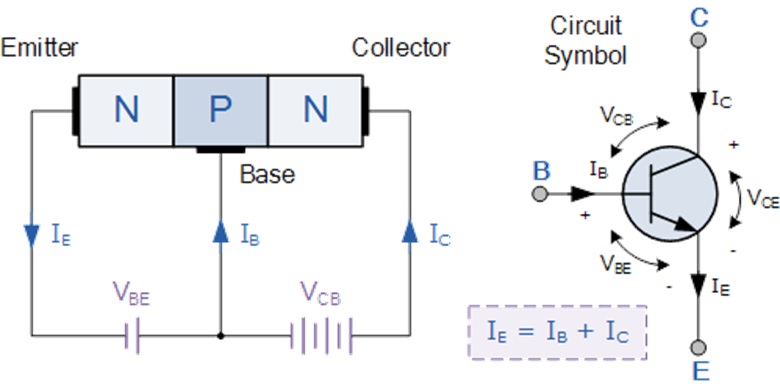
Current amplification factor in a bi junction transistor is described as the ratio of output current to its input current. In the common base configuration, current amplification factor is given as the ratio of collector current to the emitter current. The low current travels from the voltage source into the transistor base. The current at the base will turn on the transistor. Then the current is amplified and travels from the emitter of the transistor to the collector. This is how current amplification takes place in a bi junction transistor. The amplifier is a very complex circuit which is using properties of a transistor.
Here let us first write what all are given in the question.
Number of electrons entering emitter,
${{n}_{e}}={{10}^{10}}$
Time taken=${{10}^{-6}}s$
Therefore the current in the emitter is
${{I}_{e}}=\dfrac{{{q}_{e}}\times {{n}_{e}}}{t}$
${{I}_{e}}=\dfrac{{{10}^{10}}\times 1.6\times {{10}^{-19}}}{{{10}^{-6}}}$
$=1.6mA$
Since only 2% current is lost, therefore 98% will reach the collector
So
\[\alpha =0.98\]
And also we know that
${{I}_{c}}=\alpha {{I}_{e}}$
Therefore the collector current will be,
\[\begin{align}
& =\alpha \times {{I}_{e}} \\
& =0.98\times 1.6 \\
& {{I}_{c}}\approx 1.57mA \\
\end{align}\]
Also the current amplification factor will be,
\[\beta =\dfrac{\alpha }{1-\alpha }=49\]
Hence the correct answer is given by the option A.
Note:
MOSFETs are controlled by voltage not on their own. It is used to amplify current using a MOSFET by passing the input current through a resistor there by converting the current signal to a voltage. This signal voltage is controlling the MOSFET, which gives an output signal current.
${{I}_{c}}=\alpha {{I}_{e}}$
$\alpha $ is given by how much current from the emitter reaches the collector. So using this $\alpha $ amplification factor can be found out.
Complete step by step answer:
Current amplification factor in a bi junction transistor is described as the ratio of output current to its input current. In the common base configuration, current amplification factor is given as the ratio of collector current to the emitter current. The low current travels from the voltage source into the transistor base. The current at the base will turn on the transistor. Then the current is amplified and travels from the emitter of the transistor to the collector. This is how current amplification takes place in a bi junction transistor. The amplifier is a very complex circuit which is using properties of a transistor.
Here let us first write what all are given in the question.
Number of electrons entering emitter,
${{n}_{e}}={{10}^{10}}$
Time taken=${{10}^{-6}}s$
Therefore the current in the emitter is
${{I}_{e}}=\dfrac{{{q}_{e}}\times {{n}_{e}}}{t}$
${{I}_{e}}=\dfrac{{{10}^{10}}\times 1.6\times {{10}^{-19}}}{{{10}^{-6}}}$
$=1.6mA$
Since only 2% current is lost, therefore 98% will reach the collector
So
\[\alpha =0.98\]
And also we know that
${{I}_{c}}=\alpha {{I}_{e}}$
Therefore the collector current will be,
\[\begin{align}
& =\alpha \times {{I}_{e}} \\
& =0.98\times 1.6 \\
& {{I}_{c}}\approx 1.57mA \\
\end{align}\]
Also the current amplification factor will be,
\[\beta =\dfrac{\alpha }{1-\alpha }=49\]
Hence the correct answer is given by the option A.

Note:
MOSFETs are controlled by voltage not on their own. It is used to amplify current using a MOSFET by passing the input current through a resistor there by converting the current signal to a voltage. This signal voltage is controlling the MOSFET, which gives an output signal current.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Which country did Danny Casey play for class 12 english CBSE

Give 10 examples of unisexual and bisexual flowers

Coming together federation is practiced in A India class 12 social science CBSE

Write the formula to find the shortest distance between class 12 maths CBSE




