
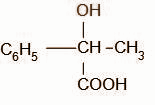
Identify D.
A.
B.
C.
D.





Answer
573.3k+ views
Hint: To answer this question, you must be familiar with reactions of various oxygen containing organic compounds. Acid chlorides are much more reactive than aldehydes, ketones and carboxylic acids.
Complete step by step solution:
In the first step acetic acid is reacted with thionyl chloride forming the product A, which is acetyl chloride.
We know that acid chlorides are more reactive than carboxylic acids, thus the compound is now susceptible to nucleophilic attack. The second reaction involves the Friedel- Craft acylation of acetyl chloride. In presence of a lewis acid, benzene ring attacks the carbonyl carbon atom and chloride is the leaving group. The product formed, B is acetophenone.
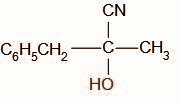
Acetophenone when reacted with hydrogen cyanide in the next step undergoes addition reaction by hydrogen cyanide over the carbon oxygen bond forming C
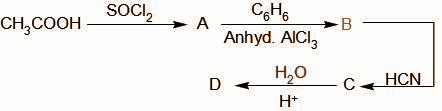
In the last step the product C hydrolyses and the cyanide group is oxidized to the carboxylic acid group. The compound D formed is,
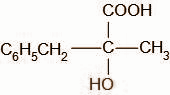
Thus, the correct option is A.
Note: Acid halides are derived from carboxylic acids by replacing the hydroxyl group in the acid by a halide. A unique property of an acid halide is its inability to form hydrogen bonds with other compounds. This absence of hydrogen bonds affects their boiling and melting point. In comparison, it can be clearly seen that the melting and boiling points of acid halides are significantly lower than those of carboxylic acids.
Acid halides are extremely reactive compounds. Each of their reactions involves the replacement of the halide group by another functional group, that might be the reacting nucleophile. All reactions of acyl halides are characterized by production of steamy acidic fumes of hydrogen halides in the first step.
Complete step by step solution:
In the first step acetic acid is reacted with thionyl chloride forming the product A, which is acetyl chloride.
We know that acid chlorides are more reactive than carboxylic acids, thus the compound is now susceptible to nucleophilic attack. The second reaction involves the Friedel- Craft acylation of acetyl chloride. In presence of a lewis acid, benzene ring attacks the carbonyl carbon atom and chloride is the leaving group. The product formed, B is acetophenone.
Acetophenone when reacted with hydrogen cyanide in the next step undergoes addition reaction by hydrogen cyanide over the carbon oxygen bond forming C
In the last step the product C hydrolyses and the cyanide group is oxidized to the carboxylic acid group. The compound D formed is,

Thus, the correct option is A.
Note: Acid halides are derived from carboxylic acids by replacing the hydroxyl group in the acid by a halide. A unique property of an acid halide is its inability to form hydrogen bonds with other compounds. This absence of hydrogen bonds affects their boiling and melting point. In comparison, it can be clearly seen that the melting and boiling points of acid halides are significantly lower than those of carboxylic acids.
Acid halides are extremely reactive compounds. Each of their reactions involves the replacement of the halide group by another functional group, that might be the reacting nucleophile. All reactions of acyl halides are characterized by production of steamy acidic fumes of hydrogen halides in the first step.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




