(i) Why is a cathode ray tube evacuated to a low pressure?
(ii) What happens if the negative potential is changed on a grid?
Answer
508.2k+ views
Hint: We know that in normal pressure conditions, air molecules are present in abundance. These molecules keep randomly moving around and cause deflection in moving particles. Also, in a grid, the negative potential supplied plays a major role.
Complete answer:
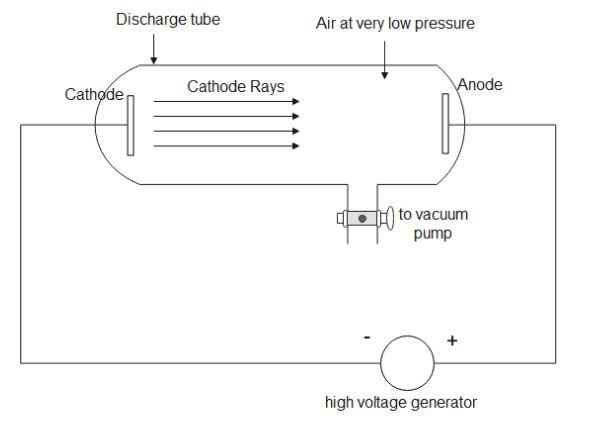
A Cathode ray tube setup consists of a discharge tube which is connected to the vacuum pump. There is an anode and a cathode inside the tube, which is connected to a high voltage generator. When this setup is switched on, a faint green luminance is produced behind the anode where the cathode rays strike.
(i) The cathode ray tube is evacuated to a low pressure so that the electrons can move freely through the tube. In the presence of normal pressure, the electrons are obstructed by the air molecules.
(ii) If the negative potential is changed on the grid, the number of electrons reaching the anode will fluctuate. This in turn will cause the number of electrons reaching the anode to change. Thus, the green luminance behind the anode will change its pattern accordingly.
Note: For better results in cathode ray experiment, an evacuated (low pressure) discharge tube is filled up with hydrogen gas. Hydrogen gas is the lightest gas (maybe the lightest element) on ionization, giving the maximum charge value to the mass ratio.
Complete answer:

A Cathode ray tube setup consists of a discharge tube which is connected to the vacuum pump. There is an anode and a cathode inside the tube, which is connected to a high voltage generator. When this setup is switched on, a faint green luminance is produced behind the anode where the cathode rays strike.
(i) The cathode ray tube is evacuated to a low pressure so that the electrons can move freely through the tube. In the presence of normal pressure, the electrons are obstructed by the air molecules.
(ii) If the negative potential is changed on the grid, the number of electrons reaching the anode will fluctuate. This in turn will cause the number of electrons reaching the anode to change. Thus, the green luminance behind the anode will change its pattern accordingly.
Note: For better results in cathode ray experiment, an evacuated (low pressure) discharge tube is filled up with hydrogen gas. Hydrogen gas is the lightest gas (maybe the lightest element) on ionization, giving the maximum charge value to the mass ratio.
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Master Class 9 Maths: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

A dentist uses a small mirror that gives a magnification class 12 physics CBSE




