What happens when salicylic acid is treated with ${(C{H_2}CO)_2}O/{H^ + }$ ?
Write chemical equations in support of your answer.
Answer
596.7k+ views
Hint:The concept of reaction of carboxylic acids will be used here in this chemical reaction. This reaction involves cleavage of $O - H$ bond when the salicylic acid is treated with ${(C{H_2}CO)_2}O/{H^ + }$.
Complete answer:
This is an acetylation reaction given in the question.
Acetylation reactions are those reactions in which acetyl groups get inserted in a compound. Here, acetyl group replaces a hydrogen atom. Acetyl group is ${H_3}C - CO$group.
When salicylic acid is treated with ${(C{H_2}CO)_2}O$ in presence of acid or ${H^ + }$ , it produces acetylsalicylic acid or aspirin.
Acetic acid is also produced as the by-product in the reaction. Salicylic acid reacts better with acetic acid than compared to when it reacts with acetic acid.
Chemical equation
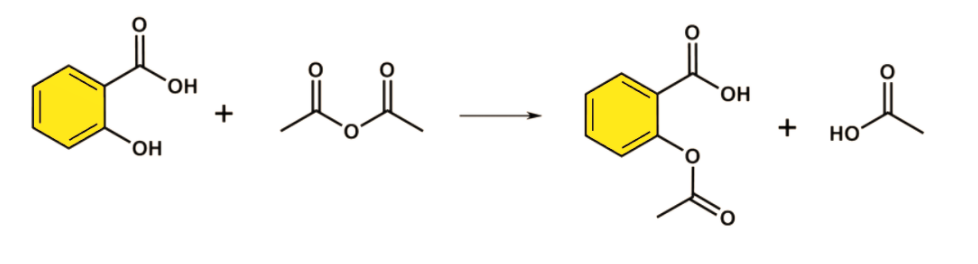
This is an irreversible reaction. This produces an adequate amount of Aspirin. Aspirin is a drug used to treat several problems.
Here, acetic anhydride is used because reaction with acetic anhydride is not reversible, so the reaction can proceed in forward reaction only. Otherwise, it might get difficult to proceed the reaction after it attains equilibrium. The reaction can be done with acetic acid instead of acetic anhydride but the reaction is reversible when it is done with acetic acid. Then the reaction will go in reverse. This is known as hydrolysis of ester. This happens due to production of water. That’s why the yield becomes less. That’s why the reaction with acetic anhydride is necessary for industrial use.
Note:
It is important to use acetic anhydride for better yield. It also helps to proceed the reaction in a forward direction. Also remember that when an acetyl group replaces a hydrogen atom in the reaction then it is known as the Acetylation reaction.
Complete answer:
This is an acetylation reaction given in the question.
Acetylation reactions are those reactions in which acetyl groups get inserted in a compound. Here, acetyl group replaces a hydrogen atom. Acetyl group is ${H_3}C - CO$group.
When salicylic acid is treated with ${(C{H_2}CO)_2}O$ in presence of acid or ${H^ + }$ , it produces acetylsalicylic acid or aspirin.
Acetic acid is also produced as the by-product in the reaction. Salicylic acid reacts better with acetic acid than compared to when it reacts with acetic acid.
Chemical equation

This is an irreversible reaction. This produces an adequate amount of Aspirin. Aspirin is a drug used to treat several problems.
Here, acetic anhydride is used because reaction with acetic anhydride is not reversible, so the reaction can proceed in forward reaction only. Otherwise, it might get difficult to proceed the reaction after it attains equilibrium. The reaction can be done with acetic acid instead of acetic anhydride but the reaction is reversible when it is done with acetic acid. Then the reaction will go in reverse. This is known as hydrolysis of ester. This happens due to production of water. That’s why the yield becomes less. That’s why the reaction with acetic anhydride is necessary for industrial use.
Note:
It is important to use acetic anhydride for better yield. It also helps to proceed the reaction in a forward direction. Also remember that when an acetyl group replaces a hydrogen atom in the reaction then it is known as the Acetylation reaction.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 9 General Knowledge: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE

Why is the cell called the structural and functional class 12 biology CBSE

Draw ray diagrams each showing i myopic eye and ii class 12 physics CBSE




