What is the geometry of complexes having hybridization $ s{p^3} $ and $ ds{p^2} $ . Give an example of each.
Answer
534k+ views
Hint: Hybridization is the mixing of atomic orbitals with the same energy to form a new degenerate orbital. The orbital thus formed is called the hybrid orbital. Hybrid orbitals possess different geometry and energy than that of the individual atomic orbitals. $ sp\,,s{p^2}\,,s{p^3}\,,s{p^3}{d^2},{d^2}sp $ etc. are examples of few hybridised orbitals.
Complete answer:
Atomic orbitals of equal energy mix to form a hybrid orbital. Both half-filled and completely filled orbitals can take part in hybridisation. The number hybrid orbitals will be equal to the number of atomic orbitals that took part in mixing.
$ s{p^3} $ hybridization: It is formed by the mixing of one s orbital and three p orbitals of the same shell of an atom that are equal in energy. Mixing of these orbitals give $ 4 $ equivalent hybrid orbitals with $ s{p^3} $ hybridization. An $ s{p^3} $ hybridised orbital contains $ 25\% $ s character and $ 75\% $ p character. The geometry of complexes possessing $ s{p^3} $ hybridization will be tetrahedral. The four atoms bound to the central atom will be directed towards the four corners of the tetrahedron. The bond angle will be $ {109^ \circ }28' $ .
Example: $ {[Ni{(Cl)_4}]^{2 - }} $ .

$ ds{p^2} $ hybridization: Mixing of $ {d_{{x^2} - {y^2}}} $ orbital, one s orbital and two p orbitals of equivalent energies gives four equivalent $ ds{p^2} $ $ ( + 2) $ hybridised orbitals. An $ ds{p^2} $ hybridized orbital contains $ 25\% $ d character, $ 25\% $ s character and $ 50\% $ p character. The geometry of a $ ds{p^2} $ hybridised complex is square planar. The bond angle between the ligand-central atom-ligand will be $ {90^ \circ } $ .
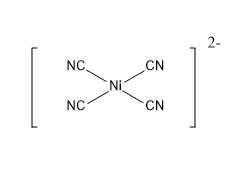
Example : $ {[Ni{(CN)_4}]^{2 - }} $
Note:
Though the central atom and its oxidation state $ ( + 2) $ are the same in both the complexes, the difference in the geometry of the complex is due to the nature of ligands. Strong ligands will form $ ds{p^2} $ complexes, while weak ligands will form $ s{p^3} $ hybridised complexes.
It should be noted that molecular orbitals and hybrid orbitals are not the same. Molecular orbitals are formed by the interaction of different atomic orbitals while hybrid orbitals are formed by the mixing of atomic orbitals of the same atom.
Complete answer:
Atomic orbitals of equal energy mix to form a hybrid orbital. Both half-filled and completely filled orbitals can take part in hybridisation. The number hybrid orbitals will be equal to the number of atomic orbitals that took part in mixing.
$ s{p^3} $ hybridization: It is formed by the mixing of one s orbital and three p orbitals of the same shell of an atom that are equal in energy. Mixing of these orbitals give $ 4 $ equivalent hybrid orbitals with $ s{p^3} $ hybridization. An $ s{p^3} $ hybridised orbital contains $ 25\% $ s character and $ 75\% $ p character. The geometry of complexes possessing $ s{p^3} $ hybridization will be tetrahedral. The four atoms bound to the central atom will be directed towards the four corners of the tetrahedron. The bond angle will be $ {109^ \circ }28' $ .
Example: $ {[Ni{(Cl)_4}]^{2 - }} $ .

$ ds{p^2} $ hybridization: Mixing of $ {d_{{x^2} - {y^2}}} $ orbital, one s orbital and two p orbitals of equivalent energies gives four equivalent $ ds{p^2} $ $ ( + 2) $ hybridised orbitals. An $ ds{p^2} $ hybridized orbital contains $ 25\% $ d character, $ 25\% $ s character and $ 50\% $ p character. The geometry of a $ ds{p^2} $ hybridised complex is square planar. The bond angle between the ligand-central atom-ligand will be $ {90^ \circ } $ .
Example : $ {[Ni{(CN)_4}]^{2 - }} $

Note:
Though the central atom and its oxidation state $ ( + 2) $ are the same in both the complexes, the difference in the geometry of the complex is due to the nature of ligands. Strong ligands will form $ ds{p^2} $ complexes, while weak ligands will form $ s{p^3} $ hybridised complexes.
It should be noted that molecular orbitals and hybrid orbitals are not the same. Molecular orbitals are formed by the interaction of different atomic orbitals while hybrid orbitals are formed by the mixing of atomic orbitals of the same atom.
Recently Updated Pages
Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Explain the structure of megasporangium class 12 biology CBSE

What are the major means of transport Explain each class 12 social science CBSE

How many chromosomes are found in human ovum a 46 b class 12 biology CBSE

The diagram of the section of a maize grain is given class 12 biology CBSE




