Explain the following with one example:
(a) Williamson’s synthesis
(b) Kolbe’s reaction
(c) Hell-volhard-zelinsky $\left( {HVZ} \right)$ reaction.
(d) Aldol reaction.
Answer
607.8k+ views
Hint: In organic reactions covalent bonds break and some new covalent bonds are formed . This making and breaking of covalent bonds usually occur in a number of steps before they are finally turned into products ; the sequential description of all the steps of the transformation into product is called the mechanism of a reaction.
Complete step by step answer:
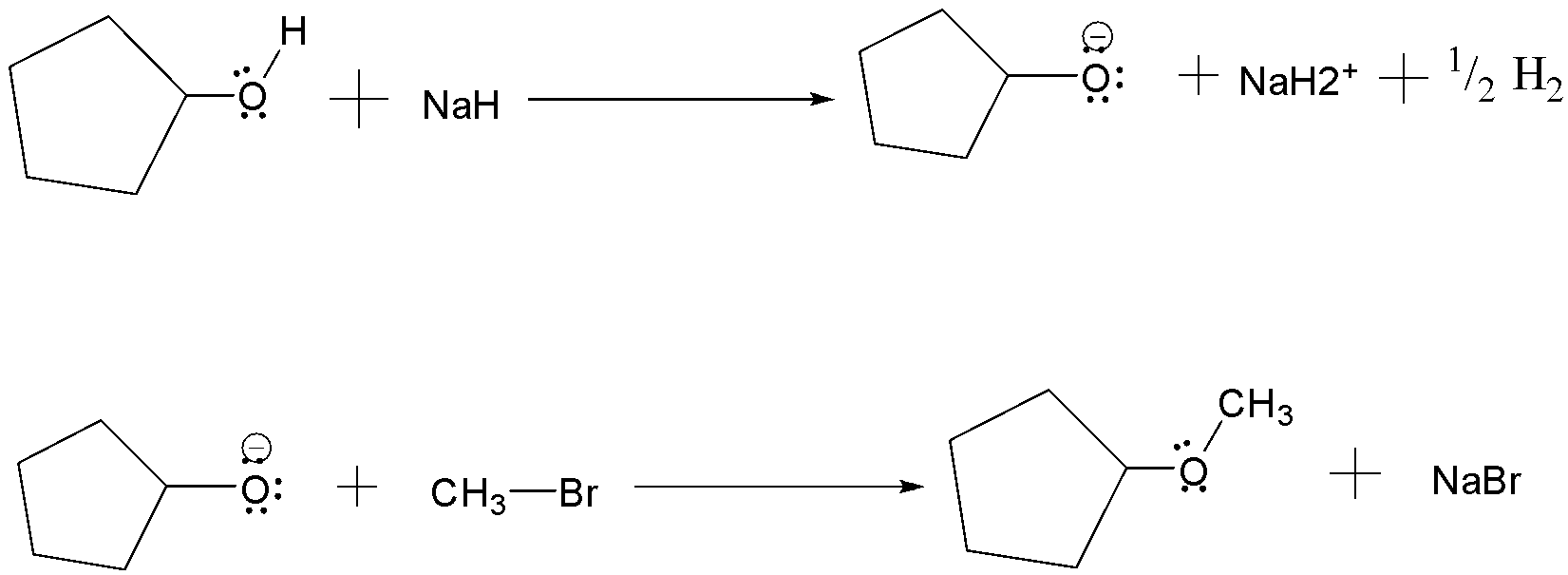
(a) Williamson’s synthesis: It is the method used for the production of ethers . It occurs by an $S{N^2}$ reaction in which a metal alkoxide displaces a halide ion from an alkyl halide. The alkoxide ion is prepared by the reaction of an alcohol with a strong base such as sodium Hydride
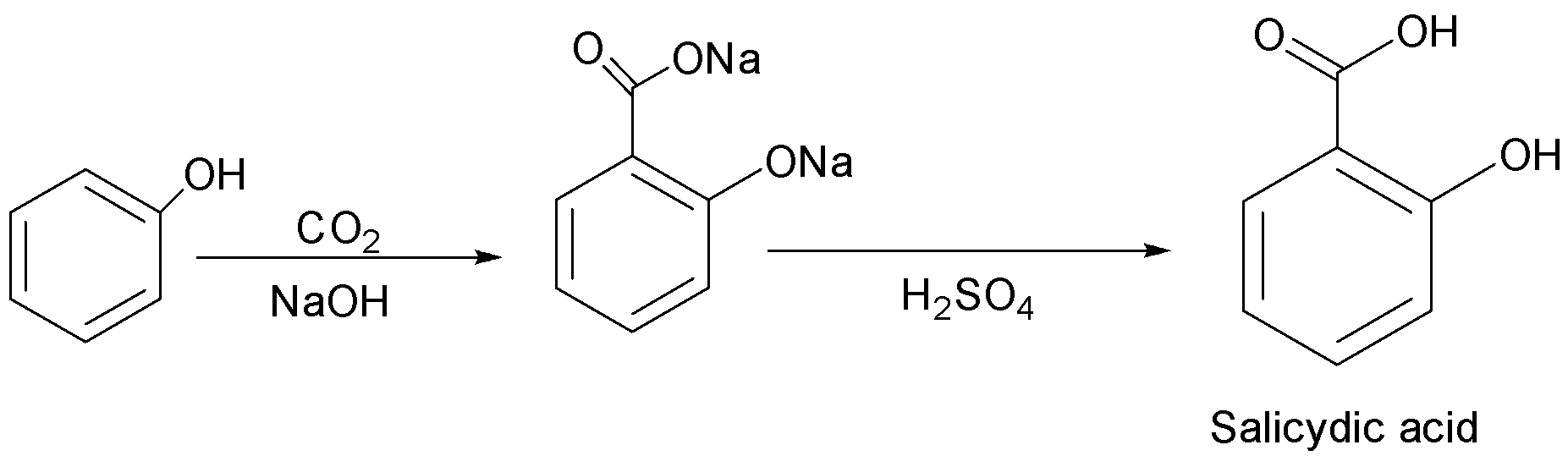
(b) Kolbe’s reaction: It is a chemical reaction that begins by first heating sodium phenoxide(the sodium salt of phenol) with carbon dioxide under pressure $\left( {100\;{\text{atm}},{{125}^ \circ }C} \right)$ , then the product is treated with sulphuric acid. The final product is an aromatic hydroxy acid which is also known as salicylic acid.
(c) Hell-volhard-zelinsky $\left( {HVZ} \right)$ reaction: When carboxylic acid reacts with chlorine or bromine using red phosphorus then halo atom is substituted at the Alpha carbon and the product obtained is mono $\alpha $ -halogenated acids. This is known as the $HVZ$ reaction.
$C{H_3}C{H_2}COOH + B{r_2}\xrightarrow{{{\text{Red}}\;{\text{P}}}}C{H_3}CHBrCOOH + HBr$
(d) Aldol reaction: The word aldol means product having both aldehyde and alcoholic group
Aldehyde having $\alpha $ -hydrogen(s) undergoes self condensation on warning with dilute or mild base to give $\beta - $ hydroxy aldehyde, called aldol (aldehyde $ + $ alcohol). This reaction is known as aldol condensation.
Note:The mechanism of a reaction is a very important part because initially we think the reactants will produce something but when we look at the mechanism we find out that actually the product is something else .
Complete step by step answer:
(a) Williamson’s synthesis: It is the method used for the production of ethers . It occurs by an $S{N^2}$ reaction in which a metal alkoxide displaces a halide ion from an alkyl halide. The alkoxide ion is prepared by the reaction of an alcohol with a strong base such as sodium Hydride

(b) Kolbe’s reaction: It is a chemical reaction that begins by first heating sodium phenoxide(the sodium salt of phenol) with carbon dioxide under pressure $\left( {100\;{\text{atm}},{{125}^ \circ }C} \right)$ , then the product is treated with sulphuric acid. The final product is an aromatic hydroxy acid which is also known as salicylic acid.

(c) Hell-volhard-zelinsky $\left( {HVZ} \right)$ reaction: When carboxylic acid reacts with chlorine or bromine using red phosphorus then halo atom is substituted at the Alpha carbon and the product obtained is mono $\alpha $ -halogenated acids. This is known as the $HVZ$ reaction.
$C{H_3}C{H_2}COOH + B{r_2}\xrightarrow{{{\text{Red}}\;{\text{P}}}}C{H_3}CHBrCOOH + HBr$
(d) Aldol reaction: The word aldol means product having both aldehyde and alcoholic group
Aldehyde having $\alpha $ -hydrogen(s) undergoes self condensation on warning with dilute or mild base to give $\beta - $ hydroxy aldehyde, called aldol (aldehyde $ + $ alcohol). This reaction is known as aldol condensation.
Note:The mechanism of a reaction is a very important part because initially we think the reactants will produce something but when we look at the mechanism we find out that actually the product is something else .
Recently Updated Pages
Master Class 12 Business Studies: Engaging Questions & Answers for Success

Master Class 12 Biology: Engaging Questions & Answers for Success

Master Class 12 Chemistry: Engaging Questions & Answers for Success

Class 12 Question and Answer - Your Ultimate Solutions Guide

Master Class 11 Social Science: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Trending doubts
Which is more stable and why class 12 chemistry CBSE

Which are the Top 10 Largest Countries of the World?

Draw a labelled sketch of the human eye class 12 physics CBSE

Differentiate between homogeneous and heterogeneous class 12 chemistry CBSE

What are the major means of transport Explain each class 12 social science CBSE

Sulphuric acid is known as the king of acids State class 12 chemistry CBSE




