
Ethylamine $({C}_{2}{H}_{5}N{H}_{2})$ can be obtained from N-ethylphthalimide on treatment with:
(A) $NaB{H}_{4}$
(B) $Ca{H}_{2}$
(C) ${H}_{2}O$
(D) $N{H}_{2}N{H}_{2}$
Answer
233.1k+ views
Hint: The Gabriel synthesis is a chemical reaction that transforms primary alkyl halides into primary amines. In this process only pure primary amines are formed and not secondary or tertiary amines.
Complete step by step answer: This synthesis is used to get primary amines from primary alkyl halides. The reaction has been generalized for applications in the alkylation of sulfonamides and imides & their deprotection in order to obtain amines. Alkylation of ammonia is quite inefficient, therefore it is substituted with phthalimide anion in the Gabriel synthesis.In this reaction N-ethylphthalimide reacts with $N{H}_{2}N{H}_{2}$ in order to form phthalhydrazide and N-ethylamine.
The following is the reaction that is included in this process.
N-ethylphthalimide is made to react with hydrazine $N{H}_{2}N{H}_{2}$. In this N-ethylphthalimide is heated with hydrazine in order to form phthalhydrazide and n-ethylamine.
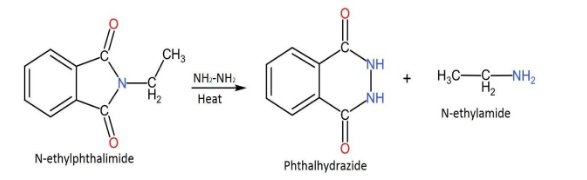
In this, the hydrazine gets attached to the carbonyl group and through a sequence of steps, the amine ends up leaving the group.
Note: N-ethyphthlamide can also be produced through a different approach also. That is with the use of an alkali KOH. Phthalimide is made to react with KOH. A good nucleophile in the form of an imide ion is formed when potassium hydroxide reacts with the phthalimide. The imide ion executes a nucleophilic substitution reaction on the alkyl halide and creates an intermediate – N-alkyl phthalimide. Then further hydrolysis of this phthalimide yields a primary alkyl amine. However, aryl amines cannot be prepared via Gabriel synthesis as aryl halides don’t undergo simple nucleophilic substitution.
Complete step by step answer: This synthesis is used to get primary amines from primary alkyl halides. The reaction has been generalized for applications in the alkylation of sulfonamides and imides & their deprotection in order to obtain amines. Alkylation of ammonia is quite inefficient, therefore it is substituted with phthalimide anion in the Gabriel synthesis.In this reaction N-ethylphthalimide reacts with $N{H}_{2}N{H}_{2}$ in order to form phthalhydrazide and N-ethylamine.
The following is the reaction that is included in this process.
N-ethylphthalimide is made to react with hydrazine $N{H}_{2}N{H}_{2}$. In this N-ethylphthalimide is heated with hydrazine in order to form phthalhydrazide and n-ethylamine.

In this, the hydrazine gets attached to the carbonyl group and through a sequence of steps, the amine ends up leaving the group.
Note: N-ethyphthlamide can also be produced through a different approach also. That is with the use of an alkali KOH. Phthalimide is made to react with KOH. A good nucleophile in the form of an imide ion is formed when potassium hydroxide reacts with the phthalimide. The imide ion executes a nucleophilic substitution reaction on the alkyl halide and creates an intermediate – N-alkyl phthalimide. Then further hydrolysis of this phthalimide yields a primary alkyl amine. However, aryl amines cannot be prepared via Gabriel synthesis as aryl halides don’t undergo simple nucleophilic substitution.
Recently Updated Pages
JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

JEE Main 2023 January 24 Shift 2 Question Paper with Answer Key

Trending doubts
JEE Main 2026: Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Angle of Deviation in a Prism

Hybridisation in Chemistry – Concept, Types & Applications

How to Convert a Galvanometer into an Ammeter or Voltmeter

Understanding the Electric Field of a Uniformly Charged Ring

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions (2025-26)

Solutions Class 12 Chemistry Chapter 1 CBSE Notes - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The d and f Block Elements (2025-26)

Biomolecules Class 12 Chemistry Chapter 10 CBSE Notes - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules (2025-26)




